Cell Biology Poster Session
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Results
Cells
We used three human cancer cell lines, T24, RT4 and A431 cells, and three immortalized human urothelial cell lines, 1T-1, 1T-2, and 1T-3 cells. T24 and RT4 cells are well-known human bladder cancer cell lines, and A431 cells are a human epidermal cancer cell line. 1T-1, 1T-2, and 1T-3 cells were established in our laboratory from normal human bladder epithelium after transfection with human papilloma virus onco-proteins, E6 and E7 (Hattori and Oyasu, manuscript in preparation). All of the three cancer cell lines are tumorigenic in nude mice, but all three immortalized urothelial cell lines are non-tumorigenic.
Expression of EGF receptors and the effect of exogenous EGF on cell growth
We have already reported the expression of EGF receptors in two bladder cancer cells, T24 and RT4 (Kameyama et al., 1993). The expression of EGF receptors in three of immortalized urothelial cell lines was also analyzed (Hattori and Oyasu, manuscript in preparation). Under our experimental conditions, only a high affinity type receptors was detected by Scatchard plot analysis. The receptor number of bladder cells ranged from 0.7 x 105 to 6.7 x 105, and the dissociation constant (kd) ranged from 0.26 to 1.31 nM (Table I). A431 cells expressed extremely high number of EGF receptors (2.3 x 106) as reported elsewhere. Exogenous EGF inhibited the growth of T24 and A431, but did not affect the growth of RT4 (Table I). The growth of T24 and A431 cells was inhibited approximately 70% below control at day 4 by either 10 ng/ml or 100 ng/ml of EGF treatment (Table I). On the other hand, EGF stimulated the growth of immortalized human urothelial cell lines, strongly of 1T-1 cells (more than 200% over control at day 4), mildly of 1T-3 cells (170% over control at day 4), and marginally of 1T-2 cells (120% over control at day 4). We examined the expression of EGF receptor protein as well as the activation of the EGF receptors after EGF treatment on the cells by Western blotting. All of the cells expressed approximately 180 kDa EGF receptors, and tyrosine residues in EGF receptors in all of the cell lines were phosphorylated after EGF treatment (data not shown). We also examined the mRNA expression of EGF receptors by semi-quantitative RT-PCR. All of the cells expressed EGF receptor mRNA. The levels of EGF receptor mRNA was roughly compatible with the levels of EGF receptor protein as well as the number of functional cell surface EGF receptors (data not shown).
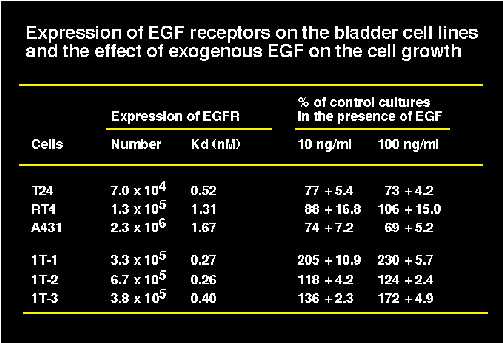 Table1
Table1Expression and activation of MAP kinases in bladder cell lines
We examined the expression and activation of MAP kinases, ERK1 and ERK2 in the cells by Western blotting. We used an affinity purified rabbit anti-MAP kinase antibody and an anti-phosphorylated MAP kinase antibody (New England Biolabs). We also used phosphorylated and non-phosphorylated 42 kDa MAP kinase (ERK2) which were obtained from New England Biolabs as a positive control. All of the cells expressed MAP kinase, however, we could not discriminate ERK1 and ERK2 under our experimental condition (Figure 1A and 1B). When we treated the cells with 100 ng/ml EGF for 15 min after 48 hour serum starvation, EGF activated the MAP kinase strongly in A431 and 1T-1 cells, and slightly in T24, 1T-2, and 1T-3 cell, but not all in RT4 cells (Figure 1C and 1D). In T24 cells, we detected the phosphorylated MAP kinase before EGF treatment. This may be due to the constitutive activation of ras-signaling by active mutation of c-Ha-ras gene in T24 cells.
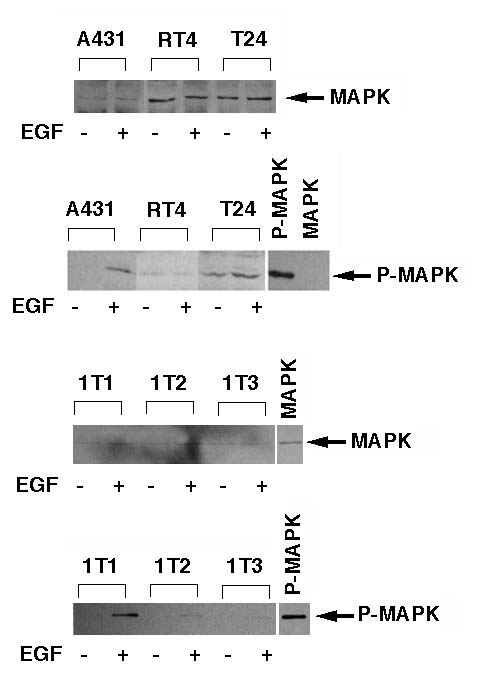 Figure1
Figure1Mobility shift assay
We examined the activation of STAT by mobility shift assay using a STAT-binding sequence, p21-SIE1 in the p21waf1 promoter region as a probe (Figure 2A). When we treated the cells with 100 ng/ml EGF for 15 min after 48 hr serum starvation, we detected the activation of STAT clearly in A431 cells and T24 cells (Figure 2B), and faintly in 1T-3 cells (Figure 2C). The mobility of the band in T24 cells was slightly faster than that in A431 and 1T-3 cells (Figure 2B and 2C).
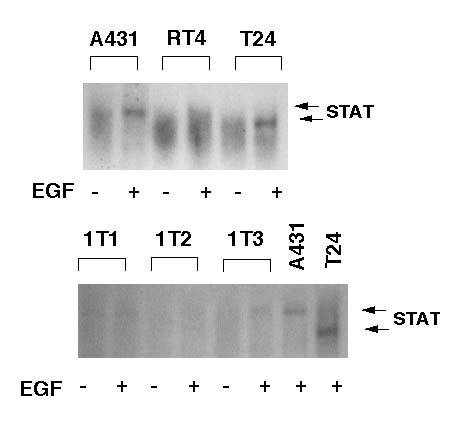 Figure 2
Figure 2Supershift assay
In order to determine the type of the activated-STAT in each cells, we performed the supershift assay. In this assay, anti-STAT1 or anti-STAT3 antibody was added to the reaction mixture before incubation with a probe. The upper band in A431 cells was recognized by the anti-STAT1 antibody, although we could not detect the supershifted slow-moving band in the gels (Figure 3). The density of the upper band was slightly reduced by the anti-STAT3 antibody, but was not erased completely (Figure 3). The lower band in T24 cells was recognized by both anti-STAT1 and anti-STAT3 antibodies, and was erased completely (Figure 3). These observations suggest that the upper band in A431 cells is probably a homodimer of STAT1, and the lower band in T24 is probably a heterodimer of STAT1 and STAT3.
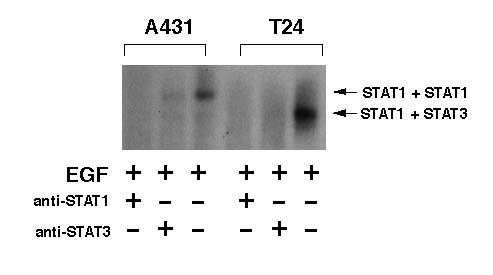 Figure 3
Figure 3Induction of p21waf1 mRNA by EGF treatment in bladder cells
We examined the expression of p21waf1 mRNA in the cells by Northern blot analysis. The mRNA expression of p21waf1 was induced after treatment with 100 ng/ml of EGF for 4 hours in A431 and T24 cells but not in RT4 cells (Figure 4A). The basal level expression of p21waf1 mRNA in immortalized urothelial cells were higher than that in cancer cells (Figure 4B). The level of p21waf1 mRNA was slightly enhanced in 1T-2 and 1T-3 cells by EGF treatment (Figure 4B). A431 (Kwok et al., 1994) and T24 cells (Kawasaki et al., 1996) were reported to have mutated p53 gene, but RT4 cells (Cooper et al., 1994) have wild type p53 gene. 1T-1, 1T-2 and 1T-3 cells were immortalized by human papilloma virus E6 and E7 onco-protein, therefore intrinsic p53 protein must be inactivate. Thus induction of p21waf1 mRNA in A431, T24, 1T-2, and 1T-3 may be mediated by p53-independent pathway.
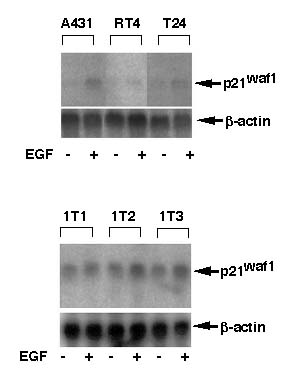 Figure 4
Figure 4Transcriptional activation of p21waf1 gene by EGF treatment
In order to determine whether EGF directly activated the transcription of p21waf1 gene in bladder cancer cells, we transfected T24 cells and RT4 cells with pGL3-WWP for luciferase assay. Fifteen hr after transfection, we treated the cells with 100 ng/ml EGF for 3 hr. Then the cell lysate was collected for luciferase assay. Luciferase activity was standardized by the protein content in the cell lysate as described in the materials and methods. EGF clearly activated the p21waf1 promoter in T24 cells, but did not in RT4 cells (Figure 5).
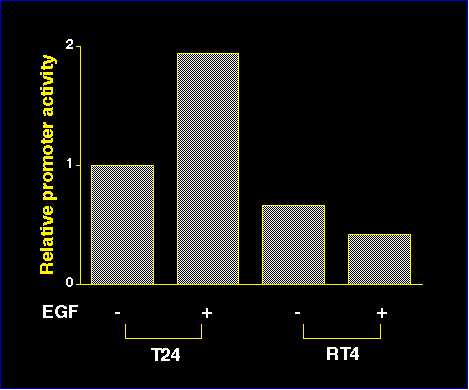 Figure 5
Figure 5| <= Materials & Methods | RESULTS | Discussion & Conclussions => |
| Discussion Board | Next Page | Your Poster Session |