Invited Symposium: SERCA-Type of Calcium Pumps and Phospholamban
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Introduction
The catalytic and transport cycle of SERCA ATPases begins with cooperative and high affinity binding of two Ca2+, whereby the enzyme is shifted from an inactive to an activated state (i.e., E2 to E1 transition). The active enzyme then proceeds with utilization of one ATP through formation of a phosphorylated intermediate. The phosphorylation reaction returns the enzyme to the E2 state and produces vectorial displacement of the two bound Ca2+, followed by hydrolytic cleavage of Pi (reviews: de Meis and Vianna, 1979; Inesi et al., 1990). The entire cycle is inhibited specifically by thapsigargin, a plant derived sesquiterpene lactone (review: Inesi and Sagara, 1994). The dependence of these reactions on the structure of the enzyme provides important clues to understand the coupling mechanism of catalytic and transport functions.
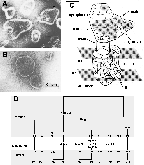
Early electron microscopic observations on sarcoplasmic reticulum vesicles (Inesi and Asai, 1968) revealed distinct granules protruding from the cytosolic surface of the membrane, and connected through a narrow stalk to a membrane bound region (Fig 1A). The granules can be removed by trypsin digestion, and correspond to the extramembranous (or cytosolic) region of the Ca2+ transport ATPase (Fig 1B). Images of ATPase molecules were subsequently derived from electron diffraction studies (Toyoshima et al., 1993), revealing an extramembranous region shaped as a bird head with a beak and a groove. The stalk and the membrane bound region were also revealed, including some detail on the orientation of membrane spanning peptide segments (Fig 1C). The ATP binding site was shown to reside within the groove of the extramembranous head (Yonekura et al., 1997).
Fig.1 - Structure of the sarcoplasmic reticulum membrane-bound ATPase A: Vesicular fragments of sarcoplasmic reticulum membrane prepared by differential centrifugation of skeletal muscle homogenate. Visualization by electron microscopy following negative staining (Scales and Inesi, 1974). Note the densely spaced granules (i.e. ATPase) with a prevalent portion protruding from the cytosolic side of the membrane, and a minor portion on the luminal side. B: Demonstration that the ATPase granules can be removed by extensive digestion with trypsin. C: Diagrammatic image of a single ATPase molecule, derived from electron diffraction studies (Toyoshima et al., 1993). D: Bidimensional diagram of the ATPase sequence (MacLennan et al., 1985) and its postulated distribution with respect to the sarcoplasmic reticulum membrane.
The distribution of the amino acid sequence (MacLennan et al., 1985) within the ATPase three dimensional structure can be approximated by examining the bidimensional diagram shown in Fig 1D. It is apparent that the small (58-107) and larger (319-763) loops are folded to yield the extramembranous region. Asp351, which is phosphorylated as an intermediate step of ATP utilization (Bastide et al., 1973; Degani and Boyer, 1973), is evidently located within or near the ATP binding groove. It is also apparent that the ten transmembrane helices (M1-10) are clustered to form the membrane bound region, connected to the head through the clustered stalk (S1-5) segments.
This article deals with experiments on localization of the Ca2+ and thapsigargin binding sites within the structure of the ATPase molecule, and the topologic relationship of these sites with the ATP binding and catalytic site.
| <= Abstract | INTRODUCTION | Materials & Methods => |
| Discussion Board | Next Page | Your Symposium |