Pharmacology & Toxicology Poster Session
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Results
While we have seen prolonged delivery of growth hormone, factor IX and B-glucuronidase in mice, preliminary experiments in dogs using our conventional APA microcapsules have not seen prolonged delivery. It was devised that the reason for the low levels of delivery was the breakage and loss of the implanted microcapsules. On day 7 after implantation only 10% of the implanted microcapsules could be recovered and none could be recovered on day 14. We have shown that other types of microcapsules can survive for more than 2 months in the dogs, but these capsules are cross-linked with Barium which has been shown to be neurotoxic.Thus, in order to develop our technology for use in larger mammals and for clinical use it will likely be necessary to create stronger microcapsules that aren't toxic. Clearly, there is a need for a method of testing the strength of new types of microcapsules in vitro before the microcapsules are implanted into more dogs.
Consequently, we have developed the osmotic pressure test as an objective, quantitative means of measuring the strength of microcapsules. Previously, strength has been measured either by subjective observation or by equating it with the thickness of the capsule wall. Subjective observation is imprecise and thus cannot detect subtle differences in microcapsule strength and can vary depending on who is looking at the capsules. While a capsule with a thicker capsule wall is generally stronger than a capsule with a thinner wall if the wall is made of the same material, the thickness of the capsule wall cannot be used as a measure of strength between different types of capsules. Futhermore, a thicker capsule wall will reduce the rate of diffusion of nutrients and wastes.
The osmotic pressure test involves exposing capsules to a series of hypotonic solutions. Upon exposure to these solutions, the capsules experience several forces which act to break the capsules, most notably the swelling of the capsule and the dilution of cross-linking calcium ions. Consequently, two samples of capsules can be compared by the percentage of intact capsules after exposure to the hypotonic solution.
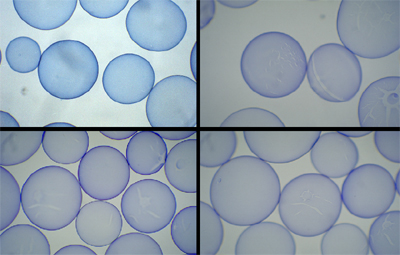 Figure 6. Sample results from an Osmotic Pressure test for one type of microcapsule. Samples are(clockwise from top left): unbroken control capsules, capsules exposed to water, capsules exposed to 0.39% serum free medium and capsules exposed to 0.78% serum free medium. Clearly, even with this small sampling of capsules, each sample can be distinguished by the percentage of intact capsules present.
Figure 6. Sample results from an Osmotic Pressure test for one type of microcapsule. Samples are(clockwise from top left): unbroken control capsules, capsules exposed to water, capsules exposed to 0.39% serum free medium and capsules exposed to 0.78% serum free medium. Clearly, even with this small sampling of capsules, each sample can be distinguished by the percentage of intact capsules present.
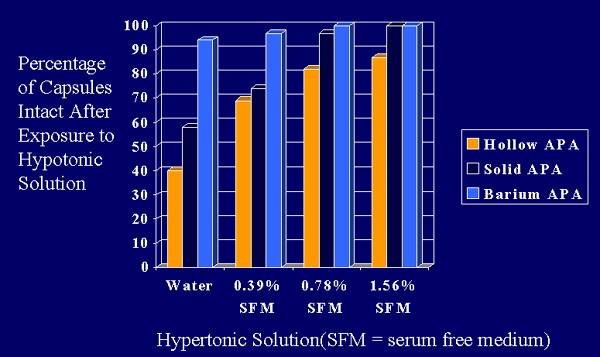 Figure 7. Results from the osmotic pressure test comparing hollow APA microcapsules, calcium cross-linked, solid APA microcapsules and barium cross-linked, solid APA microcapsules. SFM = serum free media.
Figure 7. Results from the osmotic pressure test comparing hollow APA microcapsules, calcium cross-linked, solid APA microcapsules and barium cross-linked, solid APA microcapsules. SFM = serum free media.
Clearly, the calcium cross-linked, solid APA microcapsules are stronger than the hollow APA microcapsules but weaker than the barium cross-linked APA microcapsules. While these results were predictable they serve to confirm the validity of the osmotic pressure test.
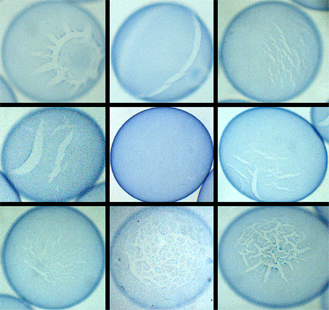 Figure 8. Pictures showing the diversity of broken capsules. An unbroken capsule is shown in the center of the figure. It is uncertain whether it is significant to call one broken capsule more broken than another or what criteria could be used to classify them.
Figure 8. Pictures showing the diversity of broken capsules. An unbroken capsule is shown in the center of the figure. It is uncertain whether it is significant to call one broken capsule more broken than another or what criteria could be used to classify them.
| <= Materials & Methods | RESULTS | Discussion & Conclussions => |
| Discussion Board | Next Page | Your Poster Session |