Invited Symposium: Na-H Exchangers and Intracellular pH Regulation
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Discussion and Conclusion
To identify amino acids important in the function of sod2, we initially characterized the physiological activity of this Na+/H+ antiporter in S. pombe. We determined the sensitivity of growth to added LiCl at various pHs in wild type and in a series of sod2 mutants. We noted that at extreme pH values (<3.5 and >5.5) growth was reduced even without external lithium. Too acidic medium is obviously harmful for these cells.
However growth also decreased at more alkaline pH's in our buffered medium. The progressive decline in the growth of S. pombe at pH> 5.0 (Fig. 2) correlates with the decrease in DpH on the plasma membrane. At an external pH of 6.5, the proton gradient is near zero (Fig. 3). Therefore the activity of DpH-driven sod2 would be reduced. It should be noted that cytosolic pH is still relatively stable among pH 3.0 to 6.5 (Fig. 3). Thus the decline of external pH rather than alkalinization of the cytoplasm, is responsible for change in DpH in more alkaline medium.
We used site-specific mutagenesis to characterize several amino acids that we suspected were critical in the activity of the enzyme. It was important to demonstrate that these mutations did not affect targeting or expression of the mutagenized proteins. We demonstrated that HA tagged variants of the mutated proteins were expressed normally in the plasma membrane of S. pombe (Fig. 4).
It should be noted that the expression of sod2 is remarkably low when governed by the natural sod2 promoter. This indicates that very few copies of the antiporter per cell are sufficient to fulfill its physiological function. The same is true for the bacterial Na+/H+ antiporter, NhaA.
The molecular analysis of sod2 revealed several amino acid residues that are critical for its function. Initially we examined the role of His residues in the function of sod2. Mutation of all the His residues of sod2 showed that only His 367 was critical for sod2 function. His 367 is one of 3 (among 8 His residues) conserved in the Na+/H+ antiporters from S. pombe, Z. rouxii, and S. cerevisiae . This supports the suggestion that it is important in sod2 function. We made several mutations of H367. The H367R, H367A and H367D mutations all resulted in a dramatic loss of the ability of yeast to grow in LiCl containing medium at pH 3.5 to 6.5 (Fig. 2). The same mutations also resulted in a complete inability to extrude Na+ in medium of acidic pH (Fig. 5). S. pombe containing the H367R, H367A mutants also showed no sod2 activity when we examined their ability to alkalinize a medium of higher pH. However the H367D mutation was active under these conditions (Fig. 6). The key difference between this experiment and the measurement of Na+ extrusion (Fig. 5) was that the measurement of Na+ extrusion was at an acidic external pH (pH 4.0) while measurement of activity of Na+-loaded yeast was in medium of pH 6.1.
This data suggests that while the HĆA replacement inactivates the antiporter, the HĆD substitution shifted the pH-optimum toward a more alkaline pH. This result suggests that His367 of sod2 protein could be regarded as a functional analog of His225 of the bacterial antiporter, NhaA. In NhaA substitution of His225 with an Asp residue also shifts the pH optimum toward a more alkaline pH. We also examined the function of three conserved Asp residues which form a '3-D motif' that is present in bacterial and yeast Na+/H+ exchangers (Fig. 1). The double mutation D266,267N inactivated sod2 completely similar to D163,164 of bacterial NhaA. In contrast, we found that cells containing the D241-sod2 mutant had almost the normal rate of 22Na efflux at acidic pH 4.0 (Figure 5C) but were unable to perform Na+-coupled proton transport at pH 6.1 (Figure 6E). We note however that the residual activity left in this mutant was unable to provide effective protection against Li+ ions in intact yeast and over a broad range of external pH (Fig. 2G). Therefore D241 appears to be an important part of sod2 activity but the antiporter still demonstrates partial activity with the DĆN mutation. It is therefore clear that this amino acid provides an important element for Na+/H+ exchanger activity by sod2 but that there is still the some sod2 activity in its absence.
On the basis of our results with the mutations of H367, D241 and D266,267 we suggest that there are some functionally significant topological invariants common for bacterial (NhaA-like) and yeast Na+/H+ antiporters (Fig. 7). These common 'Na+/H+ antiporter denominators' are: (i) a trio of Asp residues that are characteristically positioned within the neighboring transmembrane segments ('3-D motif'); and (ii) a His residue which is presumably first His downstream (of the '3-D motif') and is located at the membrane/water interface.
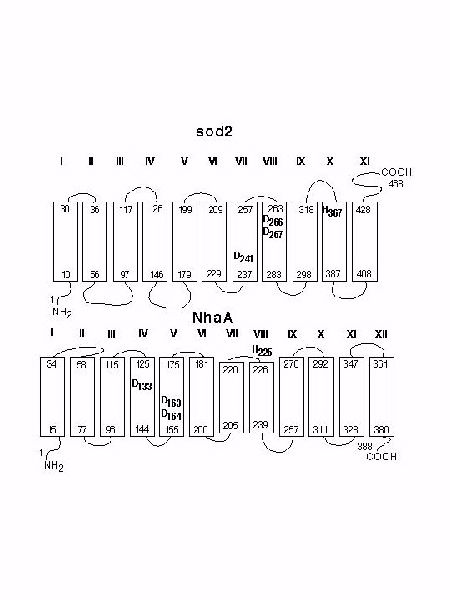 Fig. 7: Functionally important amino acid residues in sod2 and NhaA. A hypothetical model of the sod2 protein was produced using the program TopPred II. The NhaA model is based on data presented in literature.
Fig. 7: Functionally important amino acid residues in sod2 and NhaA. A hypothetical model of the sod2 protein was produced using the program TopPred II. The NhaA model is based on data presented in literature.
We speculate that conservation of isolated intramembrane Asp residues together with distances between them, reflects a general physio-chemical mechanism of cation binding by exchangers. The binding is based on coordination of the substrate cation by a crown ether-like cluster of polar amino acids, as hypothesized by Boyer. The residues involved provide their carboxylate O atoms to coordinate alkali cations and, possibly, proton in form of H3O+ ion. Modest conformational changes in such a structure could result in release of a coordinated cation at the opposite side of the membrane. Therefore, we suggest that the prokaryotic NhaA family of Na+/H+ antiporters and sod2-like yeast Na+/H+ antiporters belong to the same 'class' of exchangers exploiting a unified mechanism of cation binding.
The data presented here do not exclude the possibility that other polar residues are also involved in antiport mediated by sod2 and related exchangers. Experimental determination of the orientation and transmembrane geometry of sod2 is also highly desirable for further understanding of the structure and function of Na+/H+ exchangers. Efforts to identify additional functionally important residues and to probe the transmembrane topology of sod2 are in progress in our group.
| <= Results | DISCUSSION & CONCLUSSIONS | References => |
| Discussion Board | Next Page | Your Symposium |