Invited Symposium: Stroke/Cerebral Vasospasm
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Introduction
The etiology of cerebral vasospasm is subarachnoid blood clot. It has been suggested that hemoglobin, and possibly oxyhemoglobin specifically, is the most important spasmogenic compound released from subarachnoid erythrocytes to cause vasospasm. On the other hand, there is some controversy as to whether hemoglobin is a sufficient cause for vasospasm. Studies using magnetic resonance imaging (MRI) of the head after intracranial hemorrhage have suggested that oxyhemoglobin is reduced to low levels within hours of the hemorrhage (1). Therefore, either oxyhemoglobin could not cause delayed vasospasm or the MRI study results are erroneous. In addition, several investigators have questioned whether hemoglobin is a potent enough vasoconstrictor by itself to cause severe vasospasm (2). Aoki, et al., noted that the full vasoactive effect of erythrocyte cytosol was not due to hemoglobin alone but to a combination of hemoglobin plus a low molecular weight fraction of hemolysate (2).
These observations have led us to examine further the hypothesis that hemoglobin causes vasospasm and to think in more detail about what substances need to be present to cause vasospasm, when they need to be present, and what they need to interact with. The following work begins to investigate some of these issues.
Materials and Methods
What is the Active Component in Hemolysate Incubated In Vitro?
We first determined what substances in hemolysate from red blood cells can increase intracellular calcium ([Ca2+]i) in cerebrovascular smooth muscle cells. Smooth muscle cells were isolated by enzymatic dissociation from the basilar arteries of rats under protocols approved by the Institutional Animal Care and Use Committee. Cells were loaded with 3 µM fura 2-acetoxymethyl ester for 30 minutes and were studied using digital [Ca2+]i imaging performed by video microfluorimetry with an intensified CCD camera (Hamamatsu, Bridgewater, NJ) coupled to a computer (3,4).
The hemolysates that were studied were prepared from human blood under protocols approved by the Institutional Review Board. We tested both fresh hemolysate and hemolysate incubated in vitro for 3, 7, 14 or 21 days and also different molecular weight fractions of the hemolysates for their ability to increase [Ca2+]i in the isolated rat smooth muscle cells. The erythrocytes were obtained from whole heparinized blood by repeated washing to remove other blood components. They were either frozen immediately at -80°C or incubated in the dark at 37°C for 3, 7, 14 or 21 days. At the end of the incubation period, the red blood cells were resuspended and frozen at -80°C. For preparation of hemolysate, erythrocytes were lysed by 3 cycles of freezing at -80°C followed by thawing. We also prepared low molecular weight (LMW, <10kDa) and high molecular weight (HMW, >10kDa) fractions of the hemolysates at each time (fresh or incubated for 3, 7, 14 or 21 days) using a centrifugal ultrafiltration device (Centriprep-10, Amicon, Inc., Beverly, MA)(3,4).
Are the Substances Isolated In Vitro Active In Vivo?
Based on the results from the above experiments in vitro, we performed the following study in vivo. We randomly allocated 32 nonhuman primates to one of 4 groups. On the first day (day 0), they had cerebral angiography and placement of one of the following compounds in the subarachnoid space next to the cerebral arteries: 1. agarose (n = 8), 2. adenosine triphosphate (ATP) in agarose (n = 8), 3. autologous erythrocyte hemolysate in agarose (n = 8), or 4. purified human hemoglobin A0 in agarose (n = 8). Seven days after placement of the subarachnoid compounds (day 7), angiography was repeated and animals were euthanized. Two animals in each group were fixed by perfusion on day 7 for histopathological examination of the brain and cerebral arteries.
When Do Substances Have to be Present In Vivo to Cause Vasospasm and For How Long do They Have to be Present?
Finally, to determine when blood clot has to be present and for how long in order to cause vasospasm, nonhuman primates were randomly allocated to the groups in the following study. 24 animals had autologous clotted blood placed in the subarachnoid space bilaterally around the internal carotid, middle cerebral and anterior cerebral arteries. The diameter of each artery was measured on angiography performed on days: 0 (the day of clot placement), 1, 3, 5, 7 and 9. The subarachnoid clot was surgically removed on day 1 (4 animals), 3 (4 animals), or 5 (8 animals). Eight animals did not have the clot removed until they were euthanized. The concentrations of oxyhemoglobin and ATP were measured in the removed clots.
Measurement of Hemoglobins and Adenine Nucleotides
In all experiments, oxy- and methemoglobin concentrations of hemolysate and the HMW fraction were determined spectrophotometrically by measuring absorbance at 577 and 630 nm. High pressure liquid chromatography was used to measure concentrations of ATP, adenosine diphosphate (ADP) and adenosine monophosphate (AMP) in hemolysate (5).
Results
What is the Active Component in Hemolysate Incubated In Vitro
We found that with fresh erythrocyte hemolysate, the component that increased [Ca2+]i was contained in the LMW fraction and that the HMW fraction had no effect on [Ca2+]i. The effect of this LMW component was blocked by blockers of purinoceptors and was reproduced by ATP. Depleting ATP from fresh hemolysate also abrogated its ability to increase [Ca2+]i. Therefore, we concluded that ATP was the compound in fresh hemolysate that increases [Ca2+]i. Interestingly, pure hemoglobin did not have any effect on [Ca2+]i.
Next, we studied incubated hemolysates since in vasospasm, the blood incubates in the subarachnoid space for days before vasospasm develops. Compared to hemolysate from day 0, hemolysate from days 3, 7 or 14 produced only a transient increase in [Ca2+]i which slowly declined to the resting level within 3 minutes without a significant sustained phase. As the incubation time of aged erythrocyte hemolysate increased, the concentration necessary to increase [Ca2+]i increased. Hemolysate from day 21 produced only a minimal response even at concentrations up to 80%.
We then determined whether the activity was present in HMW or LMW fractions of incubated hemolysates. The results may be summarized as follows. The HMW fraction from any time did not evoke a response. The LMW fraction from day 3, 7 or 14 produced no response at low concentrations (<10%) and a transient response at high concentrations (> 20%) and the effect diminished with increasing incubation time. The combination of 10% LMW fraction from day 3 plus 10% HMW fraction (day 3, 7, 14 or 21) transiently increased [Ca2+]i. However, [Ca2+]i was not changed by 10% LMW fraction from day 14 plus 10% HMW fraction (from day 3 or 14). In the presence of oxyhemoglobin, [Ca2+]i was increased by 10% LMW fraction on day 3 and 7, but not by the LMW fraction from day 14 or 21. Therefore, the ability of incubated hemolysates to increase [Ca2+]i was different from the fresh hemolysate and depended on an interaction between a HMW fraction (possibly the hemoglobin in it) and a LMW fraction (not ATP, since as the results below show, there was no ATP left after incubation).
Are the Substances Isolated In Vitro Active In Vivo?
In this experiment, we tested the ability of pure ATP, pure hemoglobin or whole hemolysate to cause vasospasm in vivo. Comparisons within each group showed significant reductions in the right-sided cerebral arteries in animals exposed to ATP, hemolysate or hemoglobin (paired t-tests, p < 0.05) as can be seen in Figure 1.
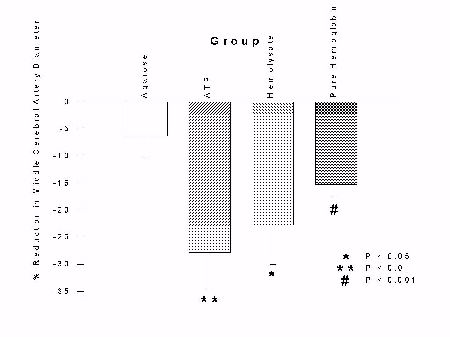 Fig. 1: Bar graphs of percent change in right middle cerebral (top), internal carotid (center) and anterior cerebral artery (bottom) diameters between day 0 and day 7, for each group. Bars represent mean � standard error. Statistically-significant reductions in diameters occurred in animals in every group except those exposed to agarose (paired t-tests).
Fig. 1: Bar graphs of percent change in right middle cerebral (top), internal carotid (center) and anterior cerebral artery (bottom) diameters between day 0 and day 7, for each group. Bars represent mean � standard error. Statistically-significant reductions in diameters occurred in animals in every group except those exposed to agarose (paired t-tests).
We examined the arteries histologically and no arteries showed histopathological changes of vasospasm. These results suggest that ATP, hemolysate and hemoglobin all can cause vasospasm this experimental paradigm but that the vasospasm is not as severe as that caused by whole blood and that it does not cause pathological changes in the arteries.
When Do Substances Have to be Present In Vivo to Cause Vasospasm and For How Long do They Have to be Present?
Monkeys without clot removal had a reduction in middle cerebral artery diameter that peaked by day 3 (33 � 13% reduction) and continued through day 9 (17 � 9% reduction). The group with clot removal on day 1 also had a reduction in middle cerebral artery diameter that peaked on day 3, followed by a significant reversal of spasm by day 5 (p < 0.01). Middle cerebral artery diameter in the day 3 clot removal group also had a peak reduction by day 3 that continued through day 5, followed by a significant reversal of spasm by day 7 (p < 0.05). Middle cerebral artery spasm in the day 5 clot removal group peaked by day 3 and continued through day 9; there was no reversal of spasm in this group. Arterial diameters remained the same as in monkeys that did not have clot removal on day 5. The results are summarized in Figures 2 and 3.
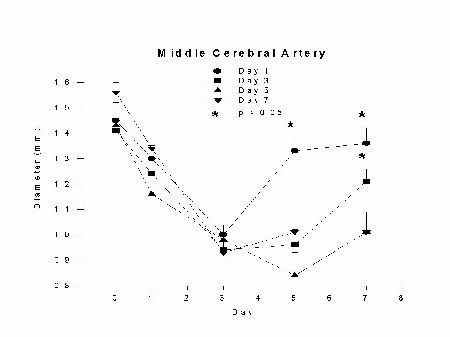 Fig. 2: Graph of middle cerebral artery diameter (mm) versus day after SAH for groups undergoing clot removal on day 1 (day 1), 3 (day 3), 5 (day 5) or no clot removal (day 7) with euthanasia on day 7. Vasospasm develops as a graded phenomenon over days and reverses significantly 4 days after clot removal on day 1 and 3 (p < 0.05, analysis of variance).
Fig. 2: Graph of middle cerebral artery diameter (mm) versus day after SAH for groups undergoing clot removal on day 1 (day 1), 3 (day 3), 5 (day 5) or no clot removal (day 7) with euthanasia on day 7. Vasospasm develops as a graded phenomenon over days and reverses significantly 4 days after clot removal on day 1 and 3 (p < 0.05, analysis of variance).
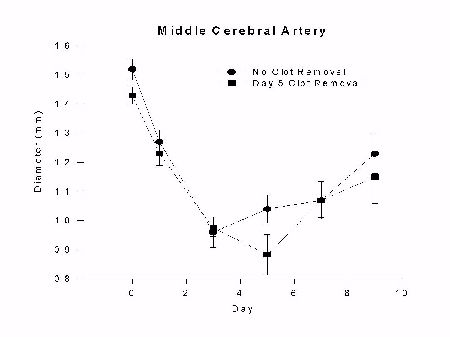 Fig. 3: Graph of middle cerebral artery diameter (mm) versus day after SAH for groups undergoing clot removal on day 5 (day 5) or no clot removal (no clot removal) with euthanasia on day 9. Vasospasm develops as a graded phenomenon over days and is not affected by clot removal on day 5.
Fig. 3: Graph of middle cerebral artery diameter (mm) versus day after SAH for groups undergoing clot removal on day 5 (day 5) or no clot removal (no clot removal) with euthanasia on day 9. Vasospasm develops as a graded phenomenon over days and is not affected by clot removal on day 5.
In summary, we have shown that removing the subarachnoid blood on or before 3 days after SAH causes vasospasm to resolve 4 days after clot removal. If the clot is removed on day 5, however, vasospasm has become temporarily irreversible and vasospasm does not resolve any sooner than it does in animals in whom the clot is not removed. Vasospasm seems to be dependent on spasmogen release within the first 3 or so days whereas by day 5 it is not.
Measurement of Hemoglobins and Adenine Nucleotides
Table 1 shows that the concentration of ATP in hemolysate incubated in vitro rapidly declined with incubation time. Hemolysate from day 0 contained a high concentration of ATP and the concentration of ATP decreased to a very low level by day 3.
Table 1: Concentration of ATP, ADP and AMP in Hemolysate From Different Incubation Times In Vitro
Time ATP (µM) ADP (µM) AMP (µM) ------------------------------------------------------------------------ Day 0 170 62 17 Day 3 0.2 0.0 1.8 Day 7 0.0 0.0 0.0
Furthermore, the same phenomenon was observed to occur in vivo. In other words, there was very little ATP present after 1 or 2 days in the clots removed from the nonhuman primates whereas hemoglobin levels remained high throughout, as seen in Table 2. The concentration of ATP left in the clots is not enough to cause significant contractions of arteries in vitro.
Table 2: Concentrations of ATP and Hemoglobins From Clots Removed From The Subarachnoid Space of Nonhuman Primates on the Indicated Days
Day Total Hemoglobin (µM) % Oxyhemoglobin ATP (µM) ----------------------------------------------------------------------- 0 430 ± 37 100 ± 1 170 1 438 ± 137 98 ± 1 .71 3 464 ± 53 89 ± 2 .41 5 412 ± 47 68 ± 8 .28 7 317 ± 69 67 ± 10 .85
Discussion and Conclusion
The findings of these studies highlight discrepancies between studies in vitro and in vivo, show that ATP cannot be an important cause of vasospasm and may be summarized as follows:
1. The [Ca2+]i imaging paradigm in vitro does not correlate with the results of experiments in vivo in the nonhuman primate model. Hemoglobin clearly causes vasospasm in vivo yet doesn't increase [Ca2+]i in vitro. This could be because the cells in vitro are not the cells that cause vasospasm in vivo, or because all of the effects of hemoglobin are indirect and mediated through perivascular nerves or binding of nitric oxide, effects that may not be observed in cells isolated in vitro.
2. Although ATP can increase [Ca2+]i in vitro, it is probably not important in vasospasm in vivo since it falls to low levels in subarachnoid clot within hours of subarachnoid hemorrhage and is not present after 3 days, for example, when the clot removal study shows that blood still needs to be present for vasospasm to persist.
3. The ability of incubated hemolysate to elevate [Ca2+]i in smooth muscle cells is due to multiple substances, including oxyhemoglobin. The substances may interact during specific times after incubation of erythrocytes in vitro.
4. ATP can cause arterial narrowing after placement in the subarachnoid space of nonhuman primates. However, levels of ATP adjacent to vasospastic arteries are not high enough to cause vasospasm. In addition, severe vasospasm with histopathological changes in the arteries equivalent to that produced by whole blood clot in the subarachnoid space of nonhuman primates was not observed. This suggests that this method of testing vasospastic substances is not a valid method of testing for substances that cause vasospasm. It should be determined if the lack of pathological changes and severe vasospasm produced by this method is because a single compound, such as ATP or hemoglobin, causes vasospasm but that placing it in agarose alters its delivery and decreases the amount of vasospasm produced or because vasospasm is a more complex, multifactorial process.
5. The clot removal study shows that vasospasm requires the presence of subarachnoid blood through day 3 but that after that, vasospasm becomes independent of the presence of subarachnoid blood clot. The reasons for this are not clear. If hemoglobin causes vasospasm, then there is some alteration in the cerebral arteries because hemoglobin is still present on day 7 yet vasospasm is resolving and does not depend on the presence of the hemoglobin which is the very substance that was postulated to cause the narrowing earlier on!
6. We feel that these studies raise important issues with regard to further investigations into vasospasm and hope you will question them to help further our understanding of vasospasm.
References
- Hayman LA, Taber KH, Ford JJ, et al (1991) Mechanisms of MR signal alteration by acute intracerebral blood: old concepts and new theories. Am J Neurorad, 12:899-907.
- Aoki T, Takenaka K, Suzuki S, et al (1994) The role of hemolysate in the facilitation of oxyhemoglobin-induced contraction in rabbit basilar arteries. J Neurosurg, 81:261-266.
- Zhang J, Weir BK, Marton LS, et al (1995) Mechanisms of hemolysate-induced [Ca++]i elevation in cerebral smooth muscle cells. Am J Physiol, 269:H1874-H1890.
- Guan YY, Weir B, Marton LS, et al (in press) Comparison of the Effects of Erythrocyte Lysate of Different Ages on [Ca2+]i in Freshly Isolated Rat Basilar Artery Smooth Muscle Cells. J Neurosurg.
- Smolenski RT, Lachno DR, Ledingham SJ, et al (1990) Determination of sixteen nucleotides, nucleosides and bases using high-performance liquid chromatography and its application to the study of purine metabolism in hearts for transplantation. J Chromatography, 527: 414-420.
| Discussion Board | Previous Page | Your Symposium |