Oxidative Stress Poster Session
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Results
Spinal cord aggregate cultures develop into round cellular spheres with an approximate diameter of 1 mm. At the LM level, control aggregates have a consistant appearance of a neuropil filled sphere comprised of a peripheral glial ring that surrounds a more centrally located neuronal cell region (Figure 1). Examples of myelination in the form of myelinated axons (profiles) can be found throughout the aggregate, but are localized mainly to the inner two thirds. At the EM level, the aggregate neuropil contains a mixture of astrocyte, oligodendrocyte and neuronal nuclei and processes as identified by Peters et al. (1991). Furthermore, typical cell to cell relationships such as synapses as well as myelinated profiles are readily identified (Figure 1C). The majority of myelinated profiles feature compact myelin lamellae surrounding healthy axons (Figure 1C).
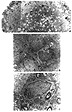 Figure 1A: Light micrograph illustrating the typical aggregate architecture and morphology. Each aggregate features a neuropil-filled sphere with scattered cell nuclei. In all cases, a peripheral ring comprised mostly of glial cell nuclei surrounds a more centrally located region containing neuronal nuclei. The large arrowhead points to an oligodendrocyte, the single long arrow points to an astrocyte and the doubled long arrows indicate a neuron. Micrometer bar = 50 µm.
Figure 1B: Electron micrograph of typical aggregate featuring a cluster of cell nuclei, most likely astrocytes (A) surrounded by healthy, compact neuropil. Micrometer bar = 2 µm.
Figure 1C: Electron micrograph featuring an oligodendrocyte cell nucleus (O) with small processes extending out to three myelinated axons (long arrows). The myelin profiles feature the characteristic compact myelin lamellae surrounding a healthy axon. Micrometer bar = 2 µm.
Figure 1A: Light micrograph illustrating the typical aggregate architecture and morphology. Each aggregate features a neuropil-filled sphere with scattered cell nuclei. In all cases, a peripheral ring comprised mostly of glial cell nuclei surrounds a more centrally located region containing neuronal nuclei. The large arrowhead points to an oligodendrocyte, the single long arrow points to an astrocyte and the doubled long arrows indicate a neuron. Micrometer bar = 50 µm.
Figure 1B: Electron micrograph of typical aggregate featuring a cluster of cell nuclei, most likely astrocytes (A) surrounded by healthy, compact neuropil. Micrometer bar = 2 µm.
Figure 1C: Electron micrograph featuring an oligodendrocyte cell nucleus (O) with small processes extending out to three myelinated axons (long arrows). The myelin profiles feature the characteristic compact myelin lamellae surrounding a healthy axon. Micrometer bar = 2 µm.
Control macrophage enriched spinal cord aggregates (Figure 2A) develop similarly to standard spinal cord aggregate cultures in that macrophages readily assimilate into the aggregate neuropil. While these aggregate cultures are smaller (approximately half the size) than standard spinal cord aggregate cultures, they retain the consistant aggregate appearance (Figure 2B) and contain numerous myelinated axons (Figure 2C).
 Figure 2A: Light micrograph of a typical aggregate culture enriched with peritoneal macrophages. These cultures retain the typical aggregate appearance of a neuropil filled sphere containing scattered cell nuclei and myelinated profiles. The peripheral glial ring still exists and many of the macrophages (long arrows) are located within and outside of this structure. The enriched cultures also contained numerous myelinated axons (small arrowhead). Micrometer bar = 50 µm.
Figure 2B: Low power EM illustrating the location of enriched macrophages (M) in the aggregate neuropil. Note the numerous examples of myelinated axons in the central portion of the aggregate (small arrowhead). Micrometer bar = 2 µm.
Figure 2C: High power EM showing the substantial presence of healthy myelinated axons in the macrophage enriched aggregate cultures. The short arrowheads point to examples of compact myelin shealths. Micrometer bar = 2 µm.
Figure 2A: Light micrograph of a typical aggregate culture enriched with peritoneal macrophages. These cultures retain the typical aggregate appearance of a neuropil filled sphere containing scattered cell nuclei and myelinated profiles. The peripheral glial ring still exists and many of the macrophages (long arrows) are located within and outside of this structure. The enriched cultures also contained numerous myelinated axons (small arrowhead). Micrometer bar = 50 µm.
Figure 2B: Low power EM illustrating the location of enriched macrophages (M) in the aggregate neuropil. Note the numerous examples of myelinated axons in the central portion of the aggregate (small arrowhead). Micrometer bar = 2 µm.
Figure 2C: High power EM showing the substantial presence of healthy myelinated axons in the macrophage enriched aggregate cultures. The short arrowheads point to examples of compact myelin shealths. Micrometer bar = 2 µm.
LM analysis of the standard spinal cord aggregates 24 hour after exposure to 5µM or 10 µM menadione, showed minor signs of neuropil disruption (Figure 3A) which included shrunken neuropil and cavities in which remnants of cells could be identified.
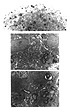 Figure 3A: Light micrograph of a spinal cord aggregate 24 hours after treatment with 10 µM of menadione. The overall architecture of these aggregates appears less organized than the control aggregates (see Figure 1A). Micrometer bar = 50 µm.
Figure 3B: Low power EM illustrating the extent of damage to the cells, neuropil and myelinated processes of the aggregate. The large arrowhead indicates a myelin profile which features a damaged neuronal process. The long arrow points to a myelinated profile that has a delamination of the myelin lamellae. The small arrowhead points to a vacuole containing cell debris. Micrometer bar = 2 µm.
Figure 3C: Electron micrograph illustrating more examples of damage to the myelinated axons. The long arrow points to a profile that has a damaged axon surrounded by a delaminated myelin sheath. The small arrowheads point to profiles with various states of neuronal and myelin damage. Micrometer bar = 2 µm.
Figure 3A: Light micrograph of a spinal cord aggregate 24 hours after treatment with 10 µM of menadione. The overall architecture of these aggregates appears less organized than the control aggregates (see Figure 1A). Micrometer bar = 50 µm.
Figure 3B: Low power EM illustrating the extent of damage to the cells, neuropil and myelinated processes of the aggregate. The large arrowhead indicates a myelin profile which features a damaged neuronal process. The long arrow points to a myelinated profile that has a delamination of the myelin lamellae. The small arrowhead points to a vacuole containing cell debris. Micrometer bar = 2 µm.
Figure 3C: Electron micrograph illustrating more examples of damage to the myelinated axons. The long arrow points to a profile that has a damaged axon surrounded by a delaminated myelin sheath. The small arrowheads point to profiles with various states of neuronal and myelin damage. Micrometer bar = 2 µm.
Most of this damage was restricted to the outer edges of the aggregates. Furthermore, the myelinated axons and more centrally located neuropil within the treated aggregates appeared unaffected. Damage to the neuropil appeared to increase in the aggregates 48 hours following menadione exposure (Figure 4A). There was also a clear concentration dependent effect. There was an increase in extracellular space, vacuoles and necrotic nuclei in all the aggregates exposed to menadione.
 Figure 4A: Light micrograph of an aggregate at 48 hours following exposure to 5 µM menadione. There is an increase in extracellular space, cavities (short arrowhead) and necrotic nuclei (long arrow). Micrometer bar = 50µm.
Figure 4B: Lower power EM illustrating the appearance of cavities in the neuropil (long arrow) within the central portions of the aggregate neuropil. Many of these areas contained the remnants of necrotic cells. Micrometer bar = 2µm.
Figure 4C: Higher power EM illustrating the extent of damage to the outer areas of the aggregate neuropil. The small arrowheads point to examples of demyelinating profiles which contain damaged and bloated axons surrounded by delaminated myelin sheaths. A central astrocyte (A) also appears to be in the process of necrosis. Micrometer bar = 2µm.
Figure 4A: Light micrograph of an aggregate at 48 hours following exposure to 5 µM menadione. There is an increase in extracellular space, cavities (short arrowhead) and necrotic nuclei (long arrow). Micrometer bar = 50µm.
Figure 4B: Lower power EM illustrating the appearance of cavities in the neuropil (long arrow) within the central portions of the aggregate neuropil. Many of these areas contained the remnants of necrotic cells. Micrometer bar = 2µm.
Figure 4C: Higher power EM illustrating the extent of damage to the outer areas of the aggregate neuropil. The small arrowheads point to examples of demyelinating profiles which contain damaged and bloated axons surrounded by delaminated myelin sheaths. A central astrocyte (A) also appears to be in the process of necrosis. Micrometer bar = 2µm.
While the deleterious effects of menadione treatment appeared more obvious 48 hours following exposure, the 96 hour samples showed no further increase in the extent of damage at the LM level. At the EM level, 24 hour treatment with menadione led to the disruption of the neuropil in the outer edges of the aggregate (compare Figure 1A and 3A) as well as the appearance of various stages of cellular necrosis. Aggregates again showed a concentration dependent increase in the presence of myelin alterations (Table 1), characterised as bloated axons surrounded by delaminated myelin sheaths (Figure 3B and C).
Time
Dose 24 Hrs. 48 Hrs. 96 Hrs.
- Mř + Mř - Mř + Mř - Mř + Mř
Control 14 17 12 21 12 18
5 µM 12 100 12 100 14 100
10 µM 26 100 27 100 33 100
20 µM 26 100 26 100 41 100
Table 1: Table outlining the extent of myelin alterations to both the standard spinal cord aggregate cultures (- Mř ) and the macrophage enriched spinal cord aggregate cultures (+ Mř ) exposed to the low doses of menadione for 24, 48 and 96 hours. All the values were calculated as percentages of the total number of myelin profiles counted. Three sections of 3 different aggregates were counted from three different experiments (n=9).
Intracellular myelin whorls were also present and many of these occurred associated with large lipid droplets (Figure 3C). Numerous extracellular whorls were located in this disrupted neuropil suggesting that these may be the remnants of myelinated axons or cell debris (Figure 3B and C). At 48 hours following exposure, the aggregates displayed further disruption to their outer architecture (Figure 4B and C). The inner areas of these aggregates however still contained healthy axons surrounded by compact myelin. At the highest dose the damage to myelin appeared more extensive (Table 1). There was no increase however in the extent of neuropil damage 96 hours following menadione exposure. LM analysis of the macrophage enriched aggregates treated with 5 µM menadione revealed severe neuropil destruction throughout the entire aggregate after 24 hours. Most of the cells within the aggregate contained shrunken nuclei and vacuolated cytoplasm (Figure 5A).
 Figure 5A: Light micrograph of a macrophage enriched spinal cord aggregate culture at 24 hours following exposure to 5µM menadione. The aggregate features severe neuropil destruction. Most of the cells within the aggregate contained shrunken nuclei and vacuolated cytoplasm (long arrows). The neuropil also featured many empty cavities corresponding to and containing the remnants of destroyed cells. Micrometer bar = 50 µm.
Figure 5B: Low power EM illustrating the extent of neuropil destruction and cell necrosis. The long arrow points to a cavity containing some cellular debris. Micrometer bar = 2 µm.
Figure 5C: Higher power EM of the extent of cell necrosis (long arrow) and damage to the myelin profiles. The short arrowheads point to myelin profiles of damaged axons surrounded by delaminated myelin sheaths. Micrometer bar = 2 µm.
Figure 5A: Light micrograph of a macrophage enriched spinal cord aggregate culture at 24 hours following exposure to 5µM menadione. The aggregate features severe neuropil destruction. Most of the cells within the aggregate contained shrunken nuclei and vacuolated cytoplasm (long arrows). The neuropil also featured many empty cavities corresponding to and containing the remnants of destroyed cells. Micrometer bar = 50 µm.
Figure 5B: Low power EM illustrating the extent of neuropil destruction and cell necrosis. The long arrow points to a cavity containing some cellular debris. Micrometer bar = 2 µm.
Figure 5C: Higher power EM of the extent of cell necrosis (long arrow) and damage to the myelin profiles. The short arrowheads point to myelin profiles of damaged axons surrounded by delaminated myelin sheaths. Micrometer bar = 2 µm.
In addition, the neuropil featured large cavities that corresponded to and contained the remnants of destroyed cells. While the added macrophages were still recognizable, other cells (neurons and oligodendrocytes) could not be readily identified. Furthermore, myelinated axons could no longer be identified in the treated cultures. The aggregates that were exposed to menadione for 48 and 96 hour exposures exhibited similar amounts of destruction. EM analysis of these macrophage enriched aggregates treated with 5 µM menadione reinforced the finding of widespread non-specific destruction of the aggregates (Figure 5B), with completely shrunken neuropil, cellular necrosis and complete demyelination and axonal destruction (Figure 5B and C). Neuropil destruction appeared to increase with time following exposure so that by 96 hours, there was a complete loss of cellular detail in the aggregates. Occassionally, examples of delaminated myelin could be identified (Figure 5C), however, in all of the macrophage enriched aggregate cultures exposed to menadione, there were no examples of normal healthy myelin (Table 1). The Nv of the individual cell types and the total cell population in the control and menadione exposed aggregates was calculated to determine whether or not one cell type was preferentially affected. In the standard aggregate cultures, exposure to different concentrations of menadione caused a decrease in the Nv in all treatment groups as time increased (Figure 6A). Individual cell populations were also analysed for Nv. Changes to Nv for oligodendrocytes and astrocytes were not significant (data not shown). With respect to the macrophage enriched aggregates that were exposed to menadione, it was not possible to readily identify individual cell types. Accordingly, only changes to the Nv of the total cell population were calculated. A significant decrease to the Nv of the total cell populations in the macrophage enriched aggregates (Figure 6B), was observed in all treatment groups.
 Figure 6A: Graph representing changes to mean numerical density of the total number of cells within spinal cord aggregate cultures following exposure to menadione. By the 96 hour time point, all treatment groups showed decreases in their Nv.
This data includes the mean values and standard error of the mean (SEM) from three separate experiments in which three separate aggregates from each treatment group were analysed (n=9).
Figure 6B: Graph representing the changes to mean numerical density (Nv) of the total number of cells within the macrophage enriched spinal cord aggregate cultures exposed to menadione. There was a significant decrease in the Nv of all the cell types exposed to all concentrations of menadione vs. control aggregates.
The data includes the mean values and standard error of the mean (SEM) from three separate experiments in which three separate aggregates from each treatment group were analysed. Statistical significance of a decrease in Nv was confirmed by ANOVA with a p = 0.0001.
Figure 6A: Graph representing changes to mean numerical density of the total number of cells within spinal cord aggregate cultures following exposure to menadione. By the 96 hour time point, all treatment groups showed decreases in their Nv.
This data includes the mean values and standard error of the mean (SEM) from three separate experiments in which three separate aggregates from each treatment group were analysed (n=9).
Figure 6B: Graph representing the changes to mean numerical density (Nv) of the total number of cells within the macrophage enriched spinal cord aggregate cultures exposed to menadione. There was a significant decrease in the Nv of all the cell types exposed to all concentrations of menadione vs. control aggregates.
The data includes the mean values and standard error of the mean (SEM) from three separate experiments in which three separate aggregates from each treatment group were analysed. Statistical significance of a decrease in Nv was confirmed by ANOVA with a p = 0.0001.
| <= Materials & Methods | RESULTS | Discussion & Conclussions => |
| Discussion Board | Next Page | Your Poster Session |