New Technology Poster Session
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Introduction
Recent studies have demonstrated that responses to single mental events (1) and brief motor tasks can be detected using fMRI. Richter et al. (2) studied sequential activation of the supplementary motor area (SMA), premotor areas and the primary motor cortex (MI) during preparatory and execution phases of a visually cued motor task. This was performed using time-resolved fMRI with relatively limited spatial coverage. As previous results indicate that the response after a single-trial task is sustained for more than 5 s, this might give opportunity to sample the elevated signal of responding areas over the whole brain. To achieve more spatial coverage, however, in the present study the sampling of BOLD signal was reduced to one instant of time per paradigm condition. Brain activation during an auditorily cued motor task was studied with functional MRI utilising a novel strategy to sample the activation signal. The sampling strategy presented in this paper aims at event-related fMRI with high spatial resolution. Activations during prepatory phase and motor task execution were studied
Materials and Methods
10 healthy volunteers (6 males, 4 females) participated in the study. The study was approved by the ethical committee of Department of Radiology of Helsinki University Central Hospital.
During the experiment the subjects kept their eyes closed. The experiment consisted of three phases each beginning with a train of four tones (duration 50 ms, ISI 250 ms). At the beginning of the preparation phase the stimulus train (cue) consisted of a random combination of two tones of different pitch (2000 Hz, 4000 Hz). At the beginning of the motor phase, a train of four similar tones (500 Hz) was presented as a go signal to execute a motor task defined by the auditory cue (2000 Hz, press left mouse button; 4000 Hz, right mouse button). The subjects were intructed to execute the motor task as soon as the go signal was over. The third phase served as control condition; after hearing a train of 250-Hz tones, the subject relaxed.
Würth 1000 ear defenders (Würth, Sankt Augustin, Germany) were used to reduce the acoustic noise from the scanner. Auditory stimuli were generated with Stim software (Neuroscan Inc., Sterling, VA, USA) running in a PC. The program controlled also the timing of image acquisition during the functional imaging. Stimuli were delivered binaurally with a homemade amplifier and loudspeaker system through plastic tubing with earplug endpieces. The system included a filter to compensate frequency distortions in the tubing.
Imaging was performed with a 1.5-T Siemens Vision system (Siemens, Erlangen, Germany). The functional images were aqcuired with a gradient-echo echo-planar sequence (TE 70 ms, TR 10920 ms, 64 slices, slice thickness 3 mm, matrix 64×64, FoV 192 mm). Transaxial slices were imaged in cranio-caudal order. The bottom slice aligned with the basal edge of the brain. The first approximately 20 slices were above the brain in order to introduce a delay to allow the BOLD response to rise. After the presentation of auditory stimuli which preceded each paradigm condition, one image volume was acquired. After the functional imaging an anatomical image set was aqcuired using a 3D-MPRAGE sequence (TE 4 ms, TR 9.7 ms, TI 20 ms, 180 slices, slice thickness 1 mm, matrix 256×256, FoV 256 mm). The subject's head was fixated with a vacuum cast in the head coil during the experiment.
Image analysis and visualisation was done with MEDx software (Sensor Systems Inc., Sterling, VA, USA). A motion correction was performed to remove subject movement effects from the data using AIR 3.0. The functional images were divided into three groups (preparation, motor execution, baseline). Pixelwise paired t-tests were calculated between preparation and baseline, and between motor execution and baseline groups. Statistical significance threshold was set to p<106 (corresponding to a Bonferroni corrected p value of 0.01 for 10000 voxels). Statistical parameter maps were co-registered with anatomical images and both of them were further transformed into Talairach coordinate space.
Results
During the preparatory period activation areas were bilaterally in the anterior cingulate , inferior frontal, medial frontal, superior frontal and precentral gyri as well as in precuneus.
The motor execution activated bilaterally inferior frontal, medial frontal, superior frontal, postcentral and precentral gyri, the cingulate motor areas, opercular cortex, inferior and superior parietal lobules and cerebellum.
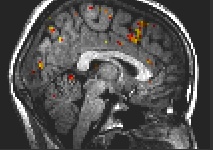 Fig. 1: Preparation, medial areas: anterior cingulate, superior frontal, precentral, postcentral gyrus, inferior parietal lobule.
Fig. 1: Preparation, medial areas: anterior cingulate, superior frontal, precentral, postcentral gyrus, inferior parietal lobule.
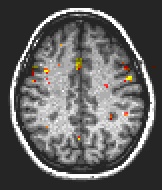 Fig. 2: Preparation: anterior cingulate, medial frontal gyrus, precuneus.
Fig. 2: Preparation: anterior cingulate, medial frontal gyrus, precuneus.
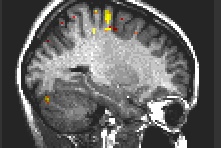 Fig. 3: Motor execution: Primary motor cortex, cerebellum.
Fig. 3: Motor execution: Primary motor cortex, cerebellum.
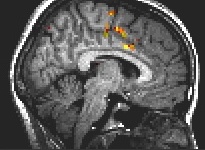 Fig. 4: Motor execution: anterior cingulate, superior frontal, precentral gyrus.
Fig. 4: Motor execution: anterior cingulate, superior frontal, precentral gyrus.
Discussion and Conclusion
We have demonstrated that it is feasible to map BOLD responses from the whole brain during a single-trial paradigm. Statistically significant activations were detected with spatial coverage extending from the most cranial cerebral areas down to the cerebellum. The test paradigm resembles those traditionally used in evoked potential studies. This allows comparison of electrophysiological data and fMRI using exactly the same experimental setup. Our investigation will be continued with a comparison with MEG and EEG data, which might reveal information about timing between the activated areas.
References
- Buckner, RL (1996) Proc. Natl. Acad. Sci. USA, 93:14878-14883.
- Richter, W (1997) NeuroReport, 8:1257-1261.
| Discussion Board | Previous Page | Your Poster Session |