Invited Symposium: Assessing DA Function using PET Techniques
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Results
Presynaptic dopaminergic function in patients with asymmetric PD
Imaging with FD, DTBZ and MP revealed a similar appearance, with asymmetric uptake and a rostrocaudal gradient, the posterior putamen being the most affected region (Fig. 1, upper). When data were expressed as a proportion of the normal values, FD uptake was increased relative to DTBZ binding (Fig. 1, lower left). In contrast, MP binding was decreased relative to DTBZ binding, particularly in patients with mild disease (Fig. 1, lower right).
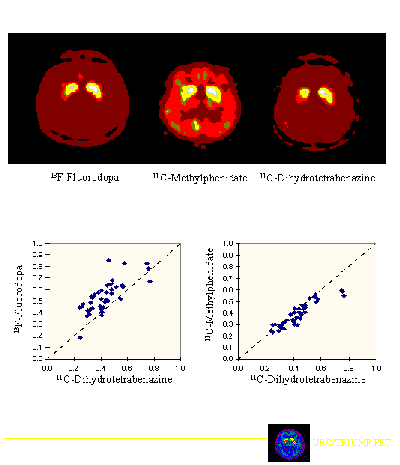 Fig. 1: Above:FD uptake, MP binding to the DAT and DTBZ binding toVMAT2 in a patient with early, asymmetric PD. Below:FD uptake (left) and MP binding (right) compared to DTBZ binding, all expressed as a proportion of the normal values. Note that FD uptake is increased relative to DTBZ binding, whereas MP binding is reduced.
Fig. 1: Above:FD uptake, MP binding to the DAT and DTBZ binding toVMAT2 in a patient with early, asymmetric PD. Below:FD uptake (left) and MP binding (right) compared to DTBZ binding, all expressed as a proportion of the normal values. Note that FD uptake is increased relative to DTBZ binding, whereas MP binding is reduced.
If one assumes that DTBZ binding will correlate directly with nerve terminal density, these findings suggest that FD uptake is relatively increased compared to the number of surviving dopamine neurons, in keeping with a compensatory upregulation of decarboxylase activity. We examined this in another manner, by determining the effect of dopamine agonist therapy with bromocriptine on FD uptake. Although the effect was not seen in all patients (scanned 18-24 h after medication withdrawal), people studied while on bromocriptine frequently had a lower FD uptake than when they were studied off agonist therapy (Fig. 2). This reduction in FD Ki while on bromocriptine is in keeping with autoreceptor-mediated downregulation of decarboxylase.
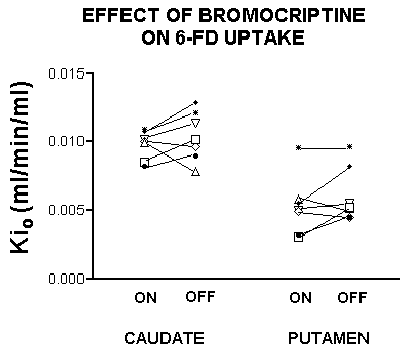 Fig. 2: FD uptake in the caudate and putamen of patients while ON or OFF bromocriptine therapy. In some patients, there is a substantial increase in FD Ki while off bromocriptine. This increase is considerably greater than that expected on the basis of scan-to-scan variability.
Fig. 2: FD uptake in the caudate and putamen of patients while ON or OFF bromocriptine therapy. In some patients, there is a substantial increase in FD Ki while off bromocriptine. This increase is considerably greater than that expected on the basis of scan-to-scan variability.
Pathophysiology of fluctuations and motor complications in PD
Even though the patients with fluctuations had a longer duration of disease, there was no significant difference in FD Ki in either the caudate or the putamen between fluctuators and patients with a stable response to levodopa (Fig. 3).
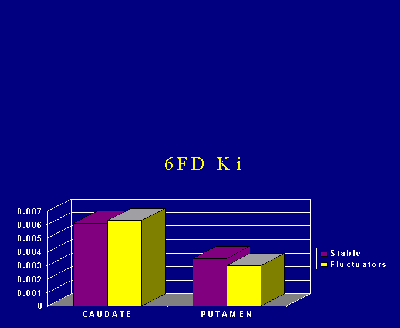 Fig. 3: FD uptake (Ki in ml/min/ml) in the caudate and putamen of patients with a stable response to levodopa and those with motor fluctuations.
Fig. 3: FD uptake (Ki in ml/min/ml) in the caudate and putamen of patients with a stable response to levodopa and those with motor fluctuations.
We have begun to examine the time course of changes in synaptic dopamine concentrations following oral levodopa. In some patients, there is a reduction in RAC binding 1 hour after levodopa (corresponding to an increase in synaptic dopamine levels), followed by a return to baseline at 4 hours (Fig. 4). We do not yet know whether there is a consistent difference between stable and fluctuating patients.
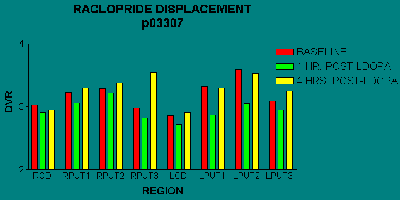 Fig. 4: RAC binding at baseline, 1 h and 4 h after levodopa in a patient with severe PD and motor fluctuations. Note that the effect is more pronounced in the putamen than the caudate, and more on the left (clinically more affected) side. Put1-3 = anterior-posterior putamen.
Fig. 4: RAC binding at baseline, 1 h and 4 h after levodopa in a patient with severe PD and motor fluctuations. Note that the effect is more pronounced in the putamen than the caudate, and more on the left (clinically more affected) side. Put1-3 = anterior-posterior putamen.
Studies of D1 and D2 binding revealed that D2 receptor binding declines in the caudate with increasing severity of disease, while D1 binding declines in the caudate as a function of disease duration and in both caudate and putamen with severity of disease. However, there were no differences between stable patients versus fluctuators or stable patients versus dyskinetics in D1 or D2 receptor binding, nor in the log ratio of D1 to D2 binding. As expected, there was an increase in D2 binding in the putamen of patients with early, untreated PD (data not shown).
Radiolabelled antagonists will bind to all affinity states of the receptor. Such single-point studies would be unable to detect shifts in the affinity of receptors for dopamine, a mechanism that has been suggested by some to underly motor complications. In order to examine the affinity of the receptor for an agonist, we have performed dose response curves for inhibition of RAC (Fig. 5) and SCH (data not shown) binding. This will be extended to see if there are differences in the ID50 between stable, fluctuating and dyskinetic patients.
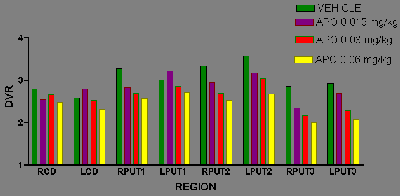 Fig. 5: RAC binding at baseline, and after increasing doses of apomorphine in a parkinsonian patient. Note that the effect is more pronounced in the putamen than the caudate, and more in the clinically more affected striatum. Put1-3 = anterior-posterior putamen.
Fig. 5: RAC binding at baseline, and after increasing doses of apomorphine in a parkinsonian patient. Note that the effect is more pronounced in the putamen than the caudate, and more in the clinically more affected striatum. Put1-3 = anterior-posterior putamen.
| <= Materials & Methods | RESULTS | Discussion & Conclussions => |
| Discussion Board | Next Page | Your Symposium |