Invited Symposium: Cardiac Ischemia Reperfusion
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Introduction
Transient exposure to dobutamine, a beta-adrenergic receptor agonist, can precondition the rat heart against post-ischemic contractile dysfunction following a prolonged ischemic period (1). The mechanism of dobutamine preconditioning is unknown. Although dobutamine preconditioning mimics the cardioprotective mechanisms of ischemic preconditioning, the signal transduction pathways resulting in protection are likely to be different, which suggests the possibility that the ultimate effectors are also different. A likely contributory mechanism of ischemic preconditioning is the attenuation of excessive, cytosolic calcium overload(2), which occurs during prolonged ischemia and initial reperfusion. While this may also play a role in dobutamine preconditioning, other mechanisms may contribute to the cardioprotection. A potential target for the deleterious effects of calcium overload is the mitochondrial fraction, which can be irreversibly disrupted due to excessive calcium accumulation during re-oxygenation. This calcium-induced injury to the mitochondria is likely to be exacerbated by loss of mitochondrial adenine nucleotides during ischemia (3) since mitochondrial nucleotides are known to have a membrane-stabilizing effect against calcium-induced swelling(3,4). Previous studies with liver tissue indicates that elevation of cytosolic cAMP levels can cause the intramitochondrial adenine nucleotide content to increase approximately 60%(4,5). Since dobutamine increases intracellular cAMP, a potential contributory mechanism of dobutamine preconditioning is preservation of mitochondrial adenine nucleotides during ischemia by first increasing the adenine nucleotide content prior to ischemia. The purpose of the present study was to determine if pre-ischemic treatment with dobutamine can increase the mitochondrial adenine nucleotide pool of the heart, thereby resulting in higher levels of mitochondrial adenine nucleotides during ischemia.
Materials and Methods
Isolated Perfused Heart.
Male Sprague-Dawley rats (200-250 grams) were heparinized, then anesthetized with sodium pentobarbital The hearts were quickly excised and placed in ice-cold 0.15 M NaCl. Following aortic cannulation, normothermic Langendorff perfusion was started with oxygenated (95% O2)/5% CO2) Krebs-Henseleit bicarbonate buffer KHB). A thin-wall , polyethylene balloon affixed to a fluid-filled piece of PE 190 tubing was inserted into the left ventricle through an incision in the left atrium. The PE tubing was connected to a pressure transducer by a three-way stopcock to allow determination of ventricular pressure and filling the balloon. The balloon was inflated with water to give an end diastolic pressure (EDP) between 5 and 10 mm of Hg. When indicated, dobutamine or vehicle (KHB) was infused by syringe pump into to perfusion buffer via a side-arm immediately above the aortic cannula. The rate of infusion of dobutamine was adjusted to give a perfusate concentration of approximately 10-8M. When indicated, global ischemia (37 °C) was produced for 40 minutes by closing a one-way stopcock inserted in the perfusion line immediately above the aorta.
Isolation of Mitochondria and Adenine Nucleotide Determination.Hearts were removed from the perfusion system, and mitochondria were isolated by differential centrifugation (6). The mitochondrial samples were extracted with perchloric acid. The neutralized extracts were analyzed for AMP, ADP, and ATP using HPLC (6). Results are expressed as total adenine nucleotide content/mg mitochondrial protein.
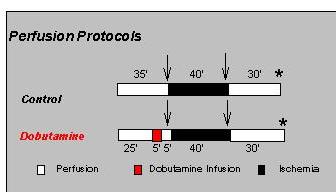
Experimental Protocol.Control and dobutamine-preconditioned hearts were studied. Hearts were preconditioned by infusion of dobutamine for 5 min followed by 5 min of a dobutamine-free perfusion as can be seen in Figure 1.
 Click to enlarge
Fig.1: Experimental protocols.
Click to enlarge
Fig.1: Experimental protocols.
Parallel studies were performed in order to measure mitochondrial adenine nucleotides (arrows, Fig. 1) prior to and at the end of a 40 min ischemic period, and to measure postischemic recovery of contractile function after ischemia (asterisks, Fig. 1).
Results
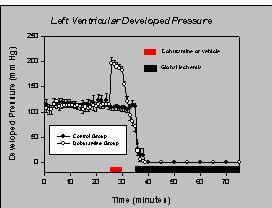
Infusion of dobutamine resulted in a 2-fold increase in developed pressure above baseline values. Upon cessation of dobutamine, pressure development decreased below baseline prior to ischemia, as seen in Figure 2.
 Click to enlarge
Fig.2: Left ventricular developed pressure during dobutamine preconditioning.
Click to enlarge
Fig.2: Left ventricular developed pressure during dobutamine preconditioning.
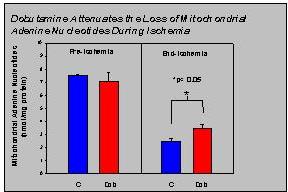
Figure 3 shows the adenine nucleotide content of mitochondria isolated from control and dobutamine-preconditioned hearts prior to ischemia and at the end of 40 min of ischemia. The pre-ischemic mitochondrial adenine nucleotide content was not significantly different between control (n=4) and dobutamine preconditioned (n=4) hearts. During ischemia, the mitochondrial adenine nucleotide content decreased approximately 65% in control hearts. In the dobutamine hearts, the mitochondrial adenine nucleotide content decreased approximately 52%. Consequently, at the end of the ischemic period, the mitochondrial adenine nucleotide content of the dobutamine preconditioned hearts (n=9) was approximately 36% higher (p<0.05) than in the control hearts as can be seen in Figure 3.
 Click to enlarge
Fig.3: Dobutamine-preconditioning attenuates the loss of mitochondrial adenine nucleotides during ischemia
Click to enlarge
Fig.3: Dobutamine-preconditioning attenuates the loss of mitochondrial adenine nucleotides during ischemia
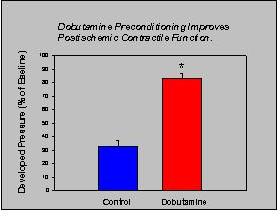
The cardioprotective effects of dobutamine preconditioning can be seen by the improved post-ischemic contractile function. Postischemic function was determined after 30 minutes of reperfusion (Fig. 1).The post-ischemic end diastolic pressure (mm Hg) was significantly lower in the dobutamine hearts compared to the control hearts (31 ± 4 vs 72 ± 3, p<.001). Moreover, the developed pressure was significantly greater (p<0.001) in the dobutamine-preconditioned hearts (n=7) compared to the control (n=10) hearts as can be seen in Figure 4.
 Click to enlarge
Fig.4: Dobutamine-preconditioning improves postischemic contractile function.
Click to enlarge
Fig.4: Dobutamine-preconditioning improves postischemic contractile function.
Discussion and Conclusion
We found that transient exposure to dobutamine did not significantly alter the total mitochondrial adenine nucleotide content of the normoxic rat heart. However, after 40 minutes of ischemia (during which time mitochondrial adenine nucleotides decrease), the mitochondrial adenine nucleotide content was greater in the dobutamine-treated hearts than the control hearts.
We conclude that dobutamine-preconditioning is associated with partial preservation of the mitochondrial adenine nucleotide pool, and this preservation is due to attenuation of nucleotide loss during prolonged ischemia.
The preservation of mitochondrial adenine nucleotides may contribute to the cardioprotective effects of dobutamine preconditioning by enhancing the tolerance of the mitochondrial fraction to calcium overload during initial reperfusion. Moreover, during reperfusion, the higher levels intramitochondrial adenine nucleotides may enhance transport of cytosolic ADP into the mitochondrial matrix for ATP synthesis (7). This may improve post-ischemic energy production for muscle contraction and promote better post-ischemic calcium homeostasis.
References
- Asimakis GK and Conti VR (1995) Preconditioning with dobutamine in the isolated heart. Life Sciences,57:177-187.
- Steenbergen C. Perlman ME, London RE and Murphy E. (1993) Mechanism of Ischemic Preconditioning: Ionic alterations Circulation Research,72:112-125.
- Asimakis GK and Sordahl LA (1981) Adenine nucleotides and energy-linked functions of cardiac mitochondria. American Journal of Physiology, 241:H672-H678.
- Pipric V, Spencer TL, and Bygrave FL (1978) Stable enhancement of calcium retention in mitochondria isolated from rat liver mitochondria after administration of glucagon to intact animals. Biochemical Journal, 176: 705-714.
- Tullson PC and Aprille JR (1987) Regulation of mitochondrial adenine nucleotide content in newborn rabbit liver. American Journal of Physiology, 253: E530-E535.
- Asimakis GK, Zwischenberger JB, Inners-McBride K, Sordahl LA and Conti VR (1992) Postischemic recovery of mitochondrial adenine nucleotides in the heart. Circulation, 85:2212-2220.
- Asimakis GK and Conti VR (1983) Myocardial ischemia: Correlation of mitochondrial adenine nucleotides and respiratory function. Journal Molecular and Cellular Cardiology, 16:439-448.
| Discussion Board | Previous Page | Your Symposium |