Invited Symposium: Neural Mechanism of Mammalian Vocalization
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Results
Vocal utterances in bats.
Bats use vocalisations for echolocation and for communication (e.g. isolation calls). The calls are produced during expiration in the larynx as in other mammalian species. The structure of the calls is basically controlled at the larynx, the filtering mechanism of the supra-laryngeal transmission pathway are relatively fixed and allow only for slight modulations of the spectral composition of the vocalisations.
The duration of echolocation calls varies between very short (some hundreds of microseconds) below ms and long in CF-FM bats (several tens of ms). A very common spectral element besides constant frequency components are frequency-modulations within a few ms. Generally, bat echolocation calls are broadband with several harmonic components. Vocalisations can be produced at high repetition rates (up to 100 per sec) and can be toggled on and off within a few milliseconds during one expiration producing call bursts. All these features of vocalisations emitted by bats require an appropriate laryngeal apparatus that is able to produce high-pitched acoustical signals that involve rapid spectral changes within the millisecond range.
Peripheral vocalisation system in the horseshoe bat.
The peripheral vocal system in the horseshoe bat, Rhinolophus rouxi, shows basic mammalian laryngeal features. Frequency control of the emitted echolocation calls is mediated by the motor branch of the superior laryngeal nerve, which originates in the rostral portion of the ventro-lateral portion of the motor nucleus, the Nucleus ambiguus. The inferior or recurrent laryngeal nerve is involved in the temporal structuring of the echolocation call components and motoneurones are located in the dorso-caudal portions of the Ncl. ambiguus. Tracer injections into physiologically characterised portions of the Ncl. ambiguus revealed a large variety of afferent and efferent connections of this nucleus (for review see: Schuller and Radtke-Schuller, 1988). To date, however, projections to the Ncl. ambiguus in the bat have only been verified anteriogradely from the cuneiform nucleus and marginal lateral parts of the periaqueductal grey (Schuller and Radtke-Schuller, unpub.). Whether a retroambigual nucleus can be distinguished in the bat and its role as structure of the final common pathway has not been investigated in the bat so far.
Where can vocalisation be elicited? Hints to structures involved in vocalisation pathway?
Electrical or pharmacological micro-stimulation is a valuable tool to determine brain structures involved in eliciting or controlling motor reactions or behavioural patterns. The conditions to evoke vocal responses are not uniform in all sensitive regions and point to differences in their functional involvement. Three main conditions could be distinguished:
a) vocalisations indistinguishable from spontaneously emitted calls couldbe elicited at very low threshold currents (less than 20 �A) without any concurrent limb or body movements; b) the stimulation elicited vocalisation that show distortions in their temporal pattern or in rare cases in their spectral composition and c) the vocalisations started after several repetitions of electrical stimulation, did not synchronise to the stimulation rhythm and persisted for some while after the electrical stimulation had been switched off.
Following the criteria of the first case, i.e. a strict one-to-one relationship between stimulus and vocalization at relatively constant response latency, five areas presumably premotor parts of the descending vocal motor system, could be delineated up to now in the brain stem:
1. the paralemniscal tegmental area (PLA) located rostrally and medially to the dorsal nucleus of the lateral lemniscus, 2. the dorsolateral parts of the mesencephalic reticular formation, corresponding to the deep mesencephalic nucleus in the rat, 3. the intermediate and deep layers of the superior colliculus (SC) which are enormously hypertrophied in the bat relative to the visual input layers, 4. the nucleus of the brachium of the inferior colliculus (NBIC) and 5. the pretectal area (AP) at the transition between superior colliculus and medial geniculate body.
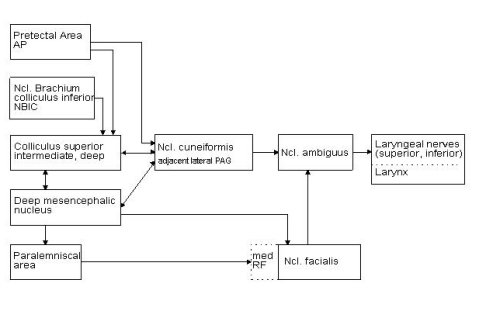 Fig.1: Vocalization can be elicited by electrical microstimulation in the brain stem areas represented in the left column. Only the most relevant connections with direct access to structures of the vocal pathway are presented.
Fig.1: Vocalization can be elicited by electrical microstimulation in the brain stem areas represented in the left column. Only the most relevant connections with direct access to structures of the vocal pathway are presented.
Shortest latencies around 25 ms were found in the PLA, whereas the latencies in the other regions could range up to 60 - 100 ms. No spectral distortions occurred in these regions and the overall spectral pattern was unaffected by the electrical stimulation parameters. These areas seem not to control directly spectral parameters but are possibly involved in gating vocal emissions under specific conditions. The activation of one of the 5 areas unilaterally is evidently sufficient to provoke vocalisation, but it is still an open question whether their activity is also a necessary condition for the emission of echolocation calls.
Stimulation in the cuneiform nucleus (cun), located latero-ventral to the periaqueductal grey and medio-ventral to the colliculi, and in the adjacent lateral periaqueductal grey region induced vocal responses with a pattern as described above under c). The vocalisations obtained at low stimulation currents (below 20 �A) were normal echolocation calls, however, the one-to-one relationship between stimulation and call was disrupted. The vocalisation also persisted for some time after stimulation had ceased and the vocal responses were accompanied by animal arousal that increased with persisting stimulation. The cuneiform nucleus relays descending vocal control information in that it has direct descending access to the laryngeal motor nucleus in the medulla and, on the other hand, receives input from four of the five regions in which vocalisation could be electrically triggered but not spectrally or temporally influenced so far.
The cuneiform nucleus presumably processes affective components of vocalisation manifested by the accompanying arousal and receives input from structures that do not influence spectro-temporal parameters of vocalisation directly. This pathway may be important for the decision whether vocalisations are uttered or not under distinct behavioural situations, but does not represent a direct vocal control interface. In this respect, the cuneiform nucleus and adjacent periaqueductal regions in the bat compare functionally to the periaqueductal grey in primates, which is considered to be an important relay station of the descending vocalisation system (J�rgens and Ploog, 1981).
Brain sites with direct influence on the spectral parameters of vocalisation are still not well defined in the bat. Areas where electrical stimulation led to distortion of vocal parameters and thus influenced the vocal output directly, were located mostly adjacent to vocal areas in lateral tegmental areas and in lateral pontine regions (Schuller and Radtke-Schuller, 1990). Spectral and temporal distortion of calls can be due either to direct influence on pre-motor or laryngeal motoneuron pools or to a temporal mismatch of respiratory and vocal control. In the latter case electrically initiated vocalisation can fall in a period of inhalation and the lack of expiratory volume can provoke the distortion of the calls. The lateral pons area yielding distorted calls and arousal also comprises the nucleus of the central acoustic tract (NCAT) with neurones tuned to the bats' constant frequency portion. It has interesting projections to putative audio-motor control structures and is considered to be a strong candidate for audio-vocal functions.
Audio-vocal structures and Dopplershift compensation (DSC) in the horseshoe bat. The possible relevance of audio-vocal structures for the Dopplershift compensation feedback system in the horseshoe bat has been investigated in several areas to different degrees. Paralemniscal tegmental area: The best investigated region is the paralemniscal area (PLA). This area appeared to be prone for an audio-vocal function in DSC as it is susceptible to electrical and pharmacological stimulation for vocalisation and embodies neurones active before and/or during vocalisation and/or active for acoustic stimulation. Most audio-vocal neurones had best frequencies in the frequency range relevant for DSC and exhibited a large activity change within a small frequency increase.
The hypothesised involvement of the paralemniscal tegmental area into the Doppler shift compensation pathway was tested with electrolytic lesions of the paralemniscal area (Pillat and Schuller, 1998). The functioning of the DSC system was neither affected by unilateral nor by bilateral lesions and only minor effects on the spontaneous vocal activity were recognised. The paralemniscal tegmental area can therefore be considered to be not necessary for the Doppler shift compensation system. However, manipulation of the gabaergic activity in the area affects vocal parameters (Schuller and Sripathi, unpub.) and these results provide evidence for modulatory influences of the PLA on vocal control.
Pretectal area: Part of the pretectal area at the transition between superior colliculus and medial geniculate body is also susceptible to electrical stimulation and vocal responses are elicited at low threshold currents. The region receives prominent afferent input from the auditory pathway, i.e. the dorsal field of the auditory cortex, the inferior colliculus (central and rostral pole nucleus) and the nucleus of the central acoustic tract. Input from non-auditory structures originate in the nucleus ruber, the deep mesencephalic nucleus and the lateral nuclei of the cerebellum. Efferent connections project back to thalamic targets (zona incerta and Ncl. reticularis thalami), to the nucleus ruber, the cuneiform nucleus and distinct areas of the pontine grey (Schuller and Radtke-Schuller, unpub.). Given the connectivity of this region, an important functional role of the pretectal area for acoustically guided behaviour in bats can be assumed, but has to be demonstrated further.
Nucleus of the brachium of the inferior colliculus: In the Ncl. of the brachium of the inferior colliculus (NBIC) in rostro-lateral position to the inferior colliculus, vocal responses can be evoked electrically at low stimulation current. This structure is of interest in the context of DSC as bilateral lesions leads to a loss of DSC behaviour, although the control of the uncompensated frequency (resting frequency) is unimpaired. Connectivity and physiological response properties of this brain region are not available in the bat.
| <= Materials & Methods | RESULTS | Discussion & Conclussions => |
| Discussion Board | Next Page | Your Symposium |