Invited Symposium: Hypertension II: Hypertension and Vascular Control
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Introduction
Role of the Renal Microvasculature in Hypertension.
Although great strides have been made in providing more effective treatments of hypertension, high blood pressure and associated cardiovascular diseases continue to be a major health problem in the United States (1). Strong support for a role of the kidney in the development of hypertension comes from renal transplantation studies in experimental animal models of hypertension and studies in human subjects (2). Additionally, in all experimental animal models of hypertension there exist a reduced ability of the kidney to excrete sodium and water.(2,3) Thus, the kidney requires a higher than normal blood pressure to maintain normal extracellular fluid volume. Hypertension could result from an enhanced renal tubular reabsorption of sodium and/or excessive renal vasoconstriction both of which impair the ability of the kidney to excrete sodium and water. Enhanced renal microvascular tone has been demonstrated in a number of hypertensive animal models and therefore, contributes to the development of hypertension.
The control of hypertension by drugs which inhibit the renin-angiotensin system points to the critical role of this system in the maintenance of hypertension (4). Furthermore, the intrarenal actions of angiotensin II are importantly involved in the long term control of arterial blood pressure (5). Two of the most important intrarenal actions of angiotensin II include constriction of the microcirculation and stimulation of tubular sodium transport (5). Chronic infusion of angiotensin II at rates too low to have an initial pressor effect cause a gradual elevation in blood pressure over several days. During the development of angiotensin II infused hypertension pressor responses to acute administration of angiotensin II are potentiated, "autopotentiation"(4). In the early stages of angiotensin II infused hypertension an increased responsiveness to angiotensin II but not norepinephrine has been demonstrated in the isolated mesenteric vasculature and aorta (4). The current studies were performed to determine if an enhanced renal microvascular tone and responsiveness to vasoconstrictors occurs during the development of angiotensin II-induced hypertension.
SPECIFIC AIMS
1.) Assess the effects of angiotensin II-induced hypertension on renal microvascular dimensions and responses to changes in perfusion pressure.
2.) Determine if angiotensin II infused hypertensive rats have enhanced renal microvascular reactivity to angiotensin II, "autopotentiation".
Materials and Methods
Induction of Angiotensin II Infused Hypertension.
Male-Sprague Dawley rats were housed separately in cages and maintained in a temperature- and light controlled room. Throughout the experiments, animals had free access to standard rat chow. All experiments were approved by the Tulane University Animal Care and Use Committee. Surgical procedures were performed with rats under pentobarbital anesthesia and with sterile procedures. Rats weighing 180 to 200 g were divided into two experimental groups: one group received sham surgery and a second group was subjected to angiotensin II infusion. Angiotensin II was infused at a continuous rate (60 ng/min) via an osmotic minipump implanted subcutaneously at the dorsum of the neck. Systolic blood pressure was measured in conscious rats by tail-cuff plethysmography to monitor the progression of hypertension. Renal microvascular studies were performed at 5-7 or 11-13 days after the start of the angiotensin II infusion.
In Vitro Perfused Juxtamedullary Preparation.
Animals were anesthetized with sodium pentobarbital and the kidney was isolated and perfused in vitro for study of the juxtamedullary vasculature. The kidney was removed from the rat, placed in an organ chamber, and continuously superfused with a warmed (37 ¯C) physiological salt solution containing 1% albumin. The kidney was perfused with a reconstituted blood (Hct = 20%). Perfusion pressure was monitored using a pressure transducer and set at 100 mmHg. Renal microvascular responses to vasoactive agonist and changes in perfusion pressure were monitored by videomicroscopy (Figure 1).
 Fig. 1:
Fig. 1:
Interlobular and afferent arteriolar diameter were measured using a digital image shearing monitor which was calibrated by a stage micrometer and measurements were reproducible within 0.5 çm. In the first series of experiments, renal perfusion pressure was lowered to 80 mmHg and the pressure-diameter relationship determined. Following the 15 min equilibration period, perfusion pressure was varied in steps form 80 to 120 to 160 mmHg. In the second series of experiments, measurements of resting afferent arteriolar diameters were made at a renal perfusion pressure of 100 mmHg. Angiotensin II (0.1-10 nM) or norepinephrine (10 nM-1çM) were administered by superfusion and diameter changes monitored for 5 minutes. Renal perfusion pressure was increased to 150 mmHg and the response to angiotensin II redetermined in animals that received angiotensin II infusion for 11-13 days.
Statistical Analysis.
All data are presented as mean Ý SEM. Difference between and within groups were analyzed by two-way ANOVA with repeated measures on one factor and Fishers least significance post hoc test. A value of &It; 0.5 was considered significant.
Results
Development of Hypertension.
Angiotensin II infusion results in a slowly developing hypertension. Systolic blood pressure increased significantly above control values by day 6 and reached 184 Ý 6 mmHg by day 12 in animals receiving angiotensin II infusion (Figure 2).
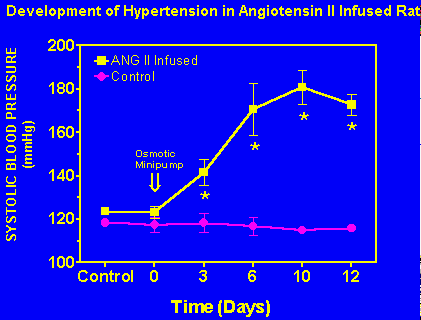 Fig. 2:
Fig. 2:
Enhanced Renal Microvascular Tone in Angiotensin II Induced Hypertension. Renal microvascular responses to increasing renal perfusion pressure were observed utilizing the in vitro juxtamedullary nephron preparation. At a renal perfusion pressure of 80 mmHg interlobular and afferent arteriolar diameters of normotensive animals averaged 28 Ý 1 and 18 Ý 1 çm; respectively. Diameters of the interlobular arteriole at renal perfusion pressures of 80, 120, and 160 mmHg were 12-20% smaller in hypertensive animals; however, arterioles from both groups responded to increases in renal perfusion pressure with similar 21-25% decreases in diameter (Figure 3).
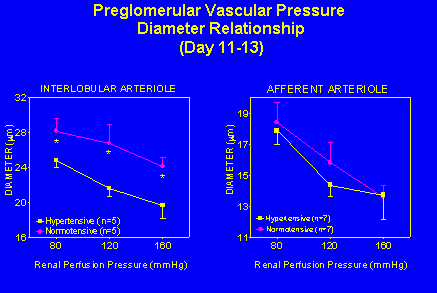 Fig. 3:
Fig. 3:
Enhanced Renal Microvascular Responsiveness to Angiotensin II in Hypertensive Animals. Angiotensin II infused hypertensive rats studied at 11-13 days had an enhanced afferent arteriolar responsiveness to angiotensin II at renal perfusion pressures of 100 and 150 mmHg (Figure 4).
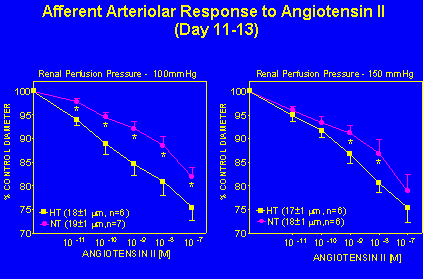 Fig. 4:
Fig. 4:
Afferent arteriolar responses to angiotensin II and norepinephrine at a renal perfusion pressure of 100 mmHg were observed in angiotensin II infused hypertensives and normotensive controls 5-7 days following osmotic minipump implantation. Angiotensin II (10 nM) decreased afferent arteriolar diameter by 27 Ý 3% in the 5-7 say hypertensive group compared to 20 Ý 2% in normotensive controls. In contrast, the afferent arteriolar response to norepinephrine was not altered after 5-7 days of angiotensin II infusion (Figure 5).
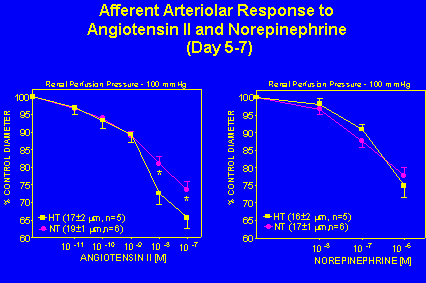 Fig. 5:
Fig. 5:
Discussion and Conclusion
SUMMARY
1.) Angiotensin II infused hypertensive rats have reduced interlobular diameters and maintained reactivity to changes in renal perfusion pressure.
2.) Afferent arterioles of hypertensive rats infused with angiotensin II for 11-13 days have an enhanced responsiveness to angiotensin II, "autopotentiation".
3.) The afferent arteriolar response to angiotensin II but not norepinephrine is enhanced following 5-7 days of angiotensin II-induced hypertension.
CONCLUSION
These data demonstrate an elevated renal microvascular tone and enhanced preglomerular reactivity which is selective for angiotensin II during the early stages of hypertension following infusion of angiotensin II. Thus, excessive renal vasoconstriction is a contributing factor to the initial defect in renal function leading to the development of hypertension.
References
- Burt VL, Whelton P, Roccella EJ, Brown C, Cutler JA, Higgens M, Horan MJ, Labarthe D (1995) Prevalence of hypertension in the US adult population: results from the third national health and nutrition examination survey. Hypertension, 25:305-313.
- Cowley AW, Roman RJ (1996) The role of the kidney in hypertension. Journal of the American Medical Association, 275:1581-1589.
- Guyton AC (1991) Blood pressure control - Special role of the kidneys and body fluids. Science, 252:1813-1816.
- Simon G, Abraham G (1995) Angiotensin II administration as an experimental model of hypertension., in Laragh JH, Brenner BM (eds): Hypertension: Pathophysiology, Diagnosis, and Management. ed. 2nd. New York, Raven Press, Ltd., pp 1423-1435.
- Mitchell KD, Navar LG (1995) Angiotensin II administration as an experimental model of hypertension., in Laragh JH, Brenner BM (eds): Hypertension: Pathophysiology, Diagnosis, and Management. ed. 2nd. New York, Raven Press, Ltd., pp 1437-1450.
| Discussion Board | Previous Page | Your Symposium |