Poster
Contents
| INABIS '98 Home Page | Your Poster Session | Related Symposia & Posters | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
(SGP-T) for Receptor Identification.
Introduction
The salivary glands have long been viewed as simple accessory organs of the digestive system. In recent years this classical view has been repeatedly challenged as these glands have demonstrated functionality in aspects of immune function, such as depressed superoxide production by neutrophils in the mouth (1), maintenance of epithelial integrity through the secretion of epithelial growth factor (EGF), which aids in maintenance and protection of the gastric mucosa (2), and alterations in nerve growth factor in response to aggression in male rats (3). Superficially, the primary function of the salivary glands appears to be the secretion of various enzymes, mucus, and water which initiate the digestive process and facilitate the unimpeded transport of ingested material along the digestive tract. It has been demonstrated that removal of the salivary glands causes a significant disruption in the ability of rats to respond to systemic shock, suggesting that the salivary glands play some homeostatic role in regulation of systemic blood pressure in response to shock inducing events such as endotoxin or antigen challenge (4, 5, 6). Along with epidermal growth factor (EGF) and nerve growth factor (NGF), which aid in maintenance of the epithelium, recently discovered gene products of Variable Coding Sequence Subclass A (VCSA1), the submandibular gland rat 1 polypeptide, the preprohormone for SGP-T, (SMR1) are also secreted. (7)
SGP-T (Thr-Asp-Ile-Phe-Glu-Gly-Gly) was originally isolated from the submandibular glands of rats based on the ability of the extract to ameliorate the hypotensive shock response due to lipopolysaccharide (LPS) (5). It was later demonstrated to reduce the contractile response of jeujenal segments isolated from ovalbumin sensitized Sprague-Dawley rats which were exposed to antigen.(6) Through the use of an alanine walk (6), it has been determined that the threonine, phenylalanine and terminal carboxyl are integral to the anti-anaphylactic properties of SGP-T. Since SGP-T does not contain any endogenous labeling site, a series of analogues containing such moieties are needed to develop probes for receptor identification of this peptide. Two such sites are investigated in the form of lysine and tyrosine, which can be labeled with biotin and iodine respectively.
Peptide Synthesis and Preparation. Peptides were synthesized by three sources: the University of Calgary Protein Synthesis Facility, Queens Protein Synthesis Facility, and University of Alberta Protein Synthesis Facility. All peptides were water soluble and dissolved in double distilled water prior to addition to the tissue baths.
Animals. Sprague-Dawley rats weighing 175-250 g were sensitized to 1 mg ovalbumin (OA) with 100 ng pertussis toxin as an adjuvant.(8) The animals were anesthetized with 0.5 ml somnotol and sacrificed by cervical distention four to six weeks following sensitization.
In vitro anaphylaxis. The procedures described previously by Mathison et al (3) were followed with slight modification. Terminal ileum was excised and 2 cm sections were mounted in 20 ml organ baths under 0.75g of tension and the isometric forces generated by OA and urecholine (URE) were measured using a Grass Force Displacement Transducer FT03. The tissues were washed several times in Krebs and allowed to equilibrate for approximately 15 minutes. Anti-anaphylactic properties of SGP-T and its analogues were determined by adding various concentrations of peptide to a bath and incubating for 10 min. Sections were then washed and exposed to 1 mg of the OA antigen. OA contractile response was measured at peak contraction. Tissues were washed and exposed to 10-5M urecholine and peak contractile response measured. Tissues were scraped of mucosa and their mass measured. Results were then normalized by calculating the ratio of OA induced contractile response to urecholine induced contractile response.
Statistical Analysis. Data are expressed as mean � standard error. The data were analyzed using one-way analysis of variance (ANOVA) and differences between groups calculated using Student's t-test for unpaired samples. Data was considered significant at P<0.05.
Results
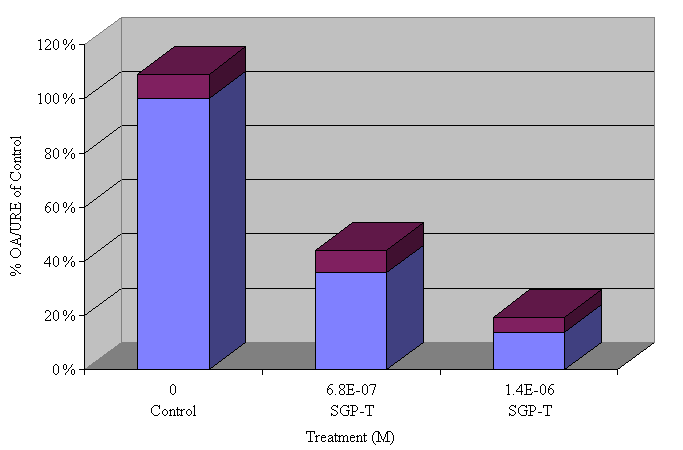
The effects of two doses of SGP-T on OA induced contraction of terminal ileum in vitro are illustrated in Figure 1. At both doses, significant reduction in antigen induced contractility was observed.
|
Figure 1. Contractile responses, expressed as a percentage
of control, following treatment with SGP-T
|
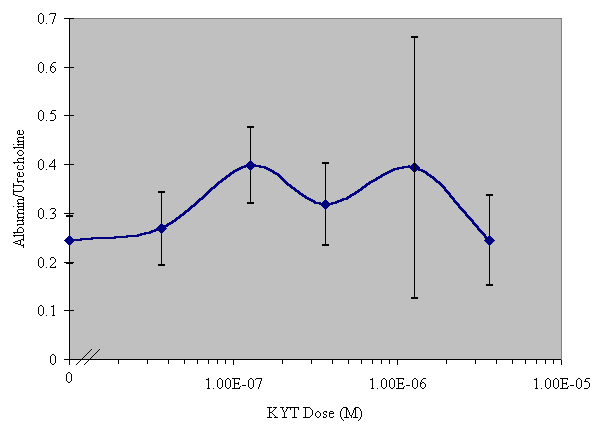
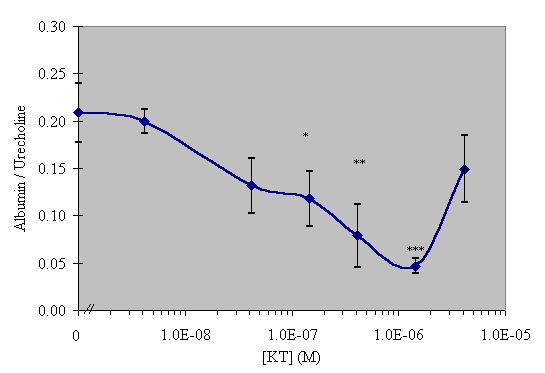
N-terminal extension of SGP-T to include a lysine and tyrosine moieties produced the molecule KYT (Lys-Tyr-Gly-Gly-Gly-Gly-Gly-Gly-Thr-Asp-Ile-Phe-Glu-Gly-Gly). This molecule contains two endogenous labeling sites in the forms of Lys and Tyr. In Vitro assay revealed that KYT had no significant biological activity in the dose range tested, as can be seen in Figure 2. However, omission of the Tyr moiety resulted in molecule KT (Lys-Gly-Gly-Gly-Gly-Gly-Gly-Thr-Asp-Ile-Phe-Glu-Gly-Gly) which retained the activity of the native SGP-T at the tested doses as is apparent in Figure 3.
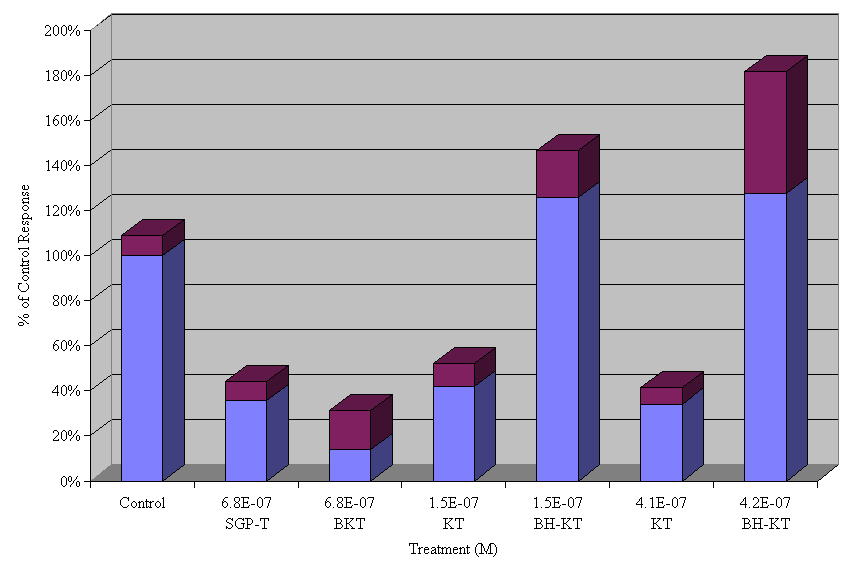
Derivatives of the biologically active KT peptide were constructed. These consisted of modification of the e-amino and/or amino terminus through use of an N-hydroxysuccinimide group, or biotinylation of these same groups. These derivatives were assayed for activity at several doses, the summarized results can be seen in Figure 4. The addition of a N-hydroxysuccinimide ester to the lysyl residue resulted in a loss of activity at all tested doses, whereas biotinylation had no adverse effect on the biological activity of the peptide.
 |
|
Figure 4. Effects of treatment with two derivatives of
KT, N-hydroxysuccinimide KT (BH-KT), and Biotinylated KT (BKT)
at various doses expressed as percent of control.
|
Discussion and Conclusion
SGP-T's anti-anaphylactic properties make it a potential target for pharmaceutical development. It appears to mediate its effects via a novel mechanism as it does not inhibit mast cell degranulation, (9) which is a key component of anaphylaxis, nor does it block the actions of histamine. (5) In order to elucidate the mechanism of action of SGP-T we are attempting to identify the cellular target of the peptide, but to this end we needed to construct biologically active analogues with appropriate labeling sites.
As can be seen in Figure 2, KYT was ineffective and similar results were obtained with the iodinated and biotinylated forms of this peptide (data not shown). The KT peptide differs from KYT only by the absence of the tyrosyl moiety and, as is shown in figure 3, retains the parent SGP-T activity. This result leads to the question, why does the presence of a tyrosine result in a loss of biological activity?
Two derivatives of KT were synthesized. The first involved the addition of an N-hydroxysuccinimide ester to nitrogen (N-terminal or e-amino group) which is identical to the hydroxylated phenyl group of tyrosine. Since the KT peptide was rendered biologically inactive after this modification (figure 4). It can be concluded that it is the direct interference of the phenol moiety which abolishes biological activity. The mechanism by which this moiety results in a loss of activity is unknown, but it may be due to a stacking interaction occurring between the two large ring structures present in the molecule (Phe, Tyr). Once this stacking occurs the peptide no longer retains a conformation compatible with its receptor.
Since biotinylation of the N-terminus resulted in a biologically active peptide (figure 4), a tool is now available for studying the receptor. The biotin, through its high affinity for avidin, allows for receptor localization by immunohistochemistry and potentially receptor isolation through the use of avidin columns.
Acknowledgments
We would like to thank the Medical Research Council of Canada for their continued financial support.
- Fu YK, Arkins S, Wang BS, Kelly KW (1991) A novel role of growth hormone and insulin-like growth factor I. Priming neutrophils for superoxide anion secretion. J Immunology 146:1602-1608.
- Li L, Yu Z, Piacik R, Hetzel DP, Rourk RM, Namiot Z, Sarosiek J, McCallum RW (1993) Effect of esophageal intraluminal mechanical and chemical stressors on salivary epidermal growth factor in humans. Am J Gastroenterology 88:1749-1755.
- Aloe L, Alleva E, Bohm, A, Levi-Montalcini R (1993) Aggressive behavior
induces release of nerve growth factor from mouse salivary gland into
the bloodstream. Proc of the Natl Acad Science USA. (1986) 83:6184-6187
- Mathison R, Befus D, Davison JS Removal of the submandibular glands increases the acute hypotensive response to endotoxin. Circ Shock 39:52-58.
- Mathison R, Befus D, Davison JS (1997) A novel submandibular gland peptide protects against endotoxic and anaphylactic shock. Am J Physiol 273: R1017-23.
- Mathison R, Davison JS, Moore G (1997) Submandibular Gland Peptide-T (Peptide-T): Modulation of Endotoxic and Anaphylactic Shock. Drug Develop Res 42:164-171
- Rougeot C, Vienet R, Cardona A, Le Doledec L, Grognet GM, Rougeon F, (1997) Targets for SMR1-pentapeptide suggest a link between circulating peptide and mineral transport. Am. J. Physiol, 273:R1309-20.
- Kosecka U, Marshall JS, Crowe SE, Bienenstock J, Perdue MH (1994) Pertusis toxin stimulates hypersensitivity and enhances nerve-mediated antigen uptake in rat intestine. Am J Physiol 267:G745-753.
| Discussion Board | Previous Page | Your Poster Session |