MATERIALS AND METHODS
The animals used in this study were twenty, 50 day-old, male
ICR rats (Nihon Crea). They had been bred with solid feed and tap
water for one week after purchase. After surgery, they had been bred
with solid and soft feed. Surgery was performed on 15 rats to cut the
masseter muscle, with the remaining 5 rats used as controls. After
they had been anaesthetized with sodium pentobarbitone (35 mg/Kg
administered interperitoneally), surgery was performed as follows:
about 4 mm deep tendon and muscle fibers that were attached to the
deep region of the right mandible, were severed while paying careful
attention to avoid damaging the nerves and blood vessels(Fig.1).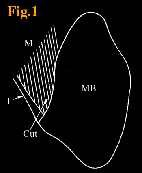 The skin was then immediately sutured. The 15 rats were divided into
3 groups of 5 rats each and thoracotomy under general anesthesia on
performed separate groups on days 3,7 and 14 after surgery. After
inserting a cannula into the left ventricle and perfused with
physiological saline solution for a short period of time, fixative (
1% paraformaldehyde and 1.5% glutaraldehyde in 0.05M phosphate buffer
pH: 7.3 ) was perfused. The mandible was then immediately removed,
fixed in the same fixative over night at 4C (celcius), and decalcified using
EDTA ( pH: 7.4 ) at 4C. After cleaning the decalcified sample in 0.1
M phosphate buffer, frontal-plane slices about 3 mm thick were cut.
According to the conventional method, slices were then embedded in
paraffin, continuous frontal-plane sections about 10 um thick cut,
and sections stained using Azan stains.
The skin was then immediately sutured. The 15 rats were divided into
3 groups of 5 rats each and thoracotomy under general anesthesia on
performed separate groups on days 3,7 and 14 after surgery. After
inserting a cannula into the left ventricle and perfused with
physiological saline solution for a short period of time, fixative (
1% paraformaldehyde and 1.5% glutaraldehyde in 0.05M phosphate buffer
pH: 7.3 ) was perfused. The mandible was then immediately removed,
fixed in the same fixative over night at 4C (celcius), and decalcified using
EDTA ( pH: 7.4 ) at 4C. After cleaning the decalcified sample in 0.1
M phosphate buffer, frontal-plane slices about 3 mm thick were cut.
According to the conventional method, slices were then embedded in
paraffin, continuous frontal-plane sections about 10 um thick cut,
and sections stained using Azan stains.
| Discussion Board
| Next Page
| Your Poster Session
|
 The skin was then immediately sutured. The 15 rats were divided into
3 groups of 5 rats each and thoracotomy under general anesthesia on
performed separate groups on days 3,7 and 14 after surgery. After
inserting a cannula into the left ventricle and perfused with
physiological saline solution for a short period of time, fixative (
1% paraformaldehyde and 1.5% glutaraldehyde in 0.05M phosphate buffer
pH: 7.3 ) was perfused. The mandible was then immediately removed,
fixed in the same fixative over night at 4C (celcius), and decalcified using
EDTA ( pH: 7.4 ) at 4C. After cleaning the decalcified sample in 0.1
M phosphate buffer, frontal-plane slices about 3 mm thick were cut.
According to the conventional method, slices were then embedded in
paraffin, continuous frontal-plane sections about 10 um thick cut,
and sections stained using Azan stains.
The skin was then immediately sutured. The 15 rats were divided into
3 groups of 5 rats each and thoracotomy under general anesthesia on
performed separate groups on days 3,7 and 14 after surgery. After
inserting a cannula into the left ventricle and perfused with
physiological saline solution for a short period of time, fixative (
1% paraformaldehyde and 1.5% glutaraldehyde in 0.05M phosphate buffer
pH: 7.3 ) was perfused. The mandible was then immediately removed,
fixed in the same fixative over night at 4C (celcius), and decalcified using
EDTA ( pH: 7.4 ) at 4C. After cleaning the decalcified sample in 0.1
M phosphate buffer, frontal-plane slices about 3 mm thick were cut.
According to the conventional method, slices were then embedded in
paraffin, continuous frontal-plane sections about 10 um thick cut,
and sections stained using Azan stains.