| INABIS '98 Home Page | Your Poster Session | Related Symposia & Posters | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Clinical examination in combination with electrophysiological recordings
and radiological imaging are widely used to evaluate the level and extent
of a spinal cord injury and to predict the degree of functional disability.
Initial ASIA limb scores (assessed by the American Spinal Injury Association
(ASIA) protocol) and both motor evoked potential (MEP) and somatosensory
evoked potential (SSEP) recordings were shown to correlate with the outcome
of ambulatory capacity after spinal cord injury (Curt and Dietz, 1997;
Curt et al., 1998).
In order to investigate further the underlying mechanisms and efficacy
of treatment approaches, animal models of spinal cord injury are of utmost
importance. The rat model can be expected as the major experimental model
to investigate new pharmacological approaches which may be of clinical
use.
The aim of the present study was to compare the outcome of spinal cord
lesion in rats and humans in order to determine whether the techniques
used in a spinal cord injury model in rats are analogous to those used
to assess spinal cord injury in man. If the rat model can be considered
to be comparable with human spinal cord injury there are essential consequences
for future therapeutical interventions, including regeneration studies.
The study design focused on the correlation between rats and humans with
respect to outcome of motor function, electrophysiological recordings,
and morphological data (high-resolution magnetic resonance imaging (MRI)
and histology) after spinal cord injury.
Materials and Methods
In rats and humans, the comparison of outcome parameters was investigated
at a time point after spinal cord injury at which a stable chronic neuropathological
condition can be expected. This time point is approximately three months
after injury for rats and at least six months after the trauma for humans.
For the animal study, 27 adult Lewis rats of either sex with spinal
cord contusion at thoracal level T8 of varying severities were used (NYU
impactor; Gruner, 1992; Constantini and Young, 1994). In the human experiments,
the patient group consisted of 54 paraplegic (45 men, 9 women, range 19
to 77 years, and 31 tetraplegic (27 men, 4 women, range 14 to 85 years)
patients.
The outcome parameters included:
Table 1: Summary of correlations of functional and anatomical parameters of rats and humans (grey), showing correlation coefficients (r-value) and p-values. The significant levels are noted for the correlation coefficients with asterisks: *p < 0.05; **p < 0.01; ***p < 0.001.
Characteristic
Vs
Characteristic
r-value

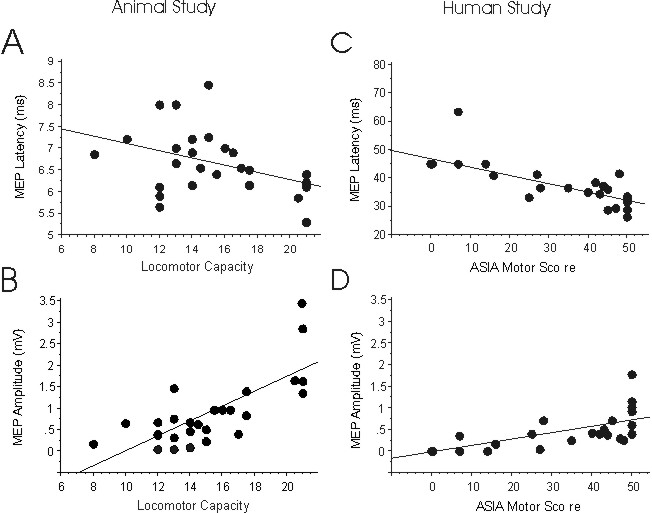
Figure 1: A-B: Scattergrams of correlations between the latency (ms)
or amplitude (mV) of MEP measurements in spinal cord injured rats at a
chronic stage as a function of open field locomotor scores of the same
animals. C-D illustrate the correlations between latency or amplitude of
MEP recordings with the ASIA lower limb motor scores of human patients
with spinal cord injury. The correlations between these functional tests
revealed a significant relationship in the animal model and in patients.
Blank plots illustrate estimated values substituted for missing
potentials.
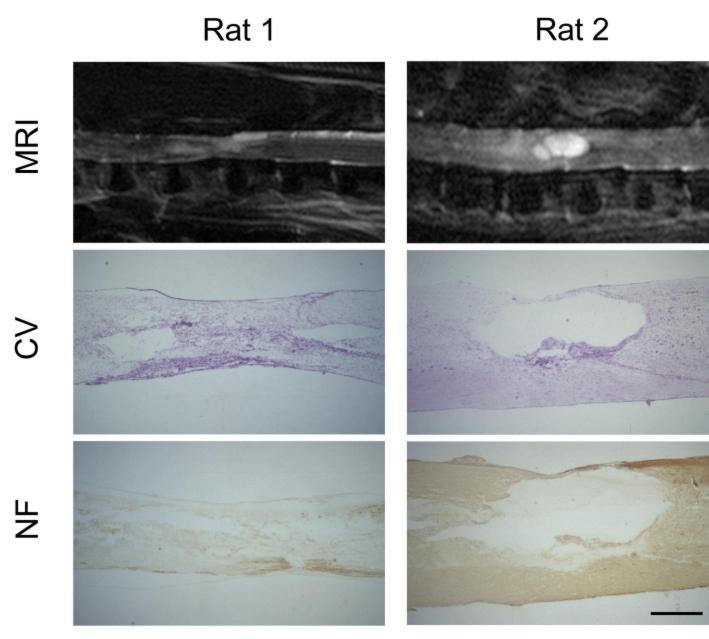
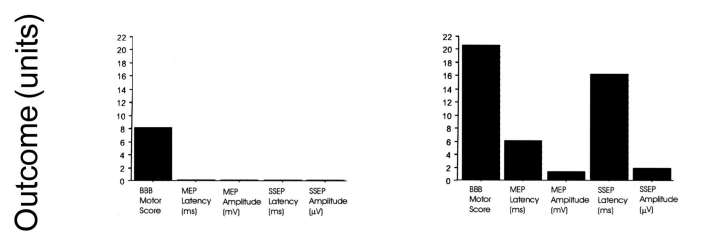
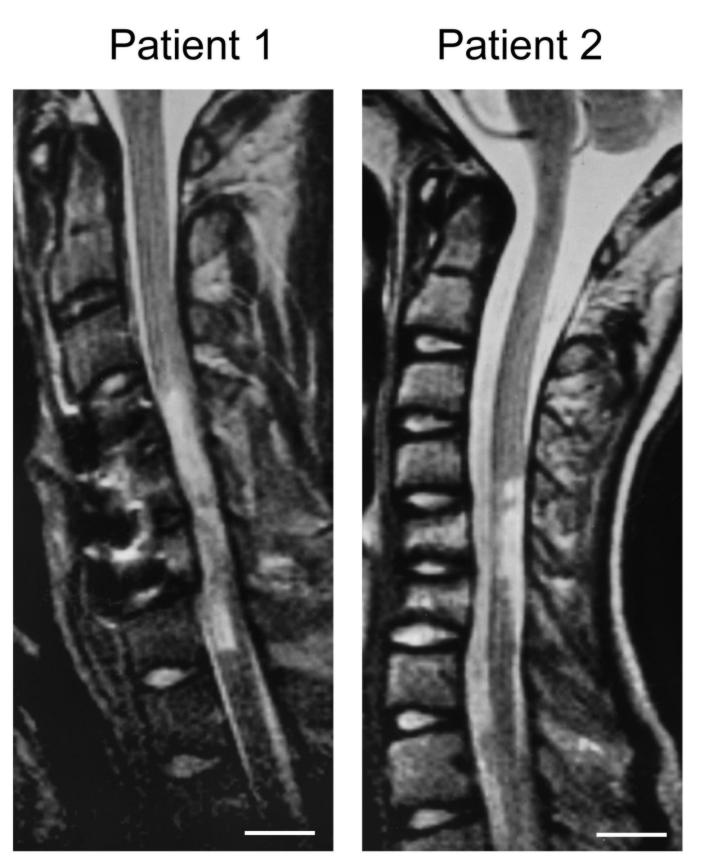
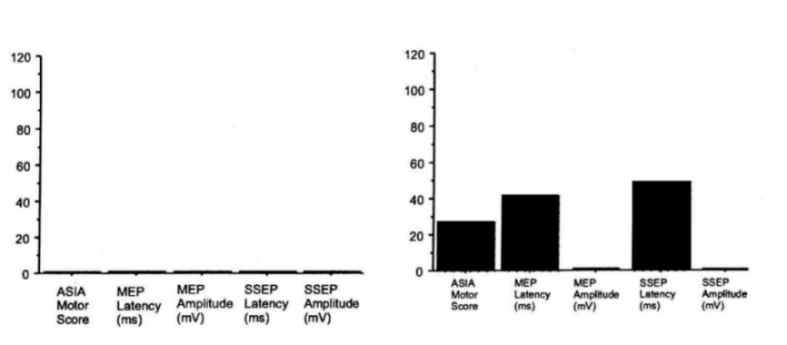
Figure 2: Comparison between morphological and functional data in
two rats (Rat 1: low locomotor capacity; Rat 2: high locomotor capacity)
and two human patients (Patient 1: low ASIA lower limb motor score; Patient
2: higher ASIA lower limb motor score) with spinal cord injury at a chronic
stage. MRI: T2-weighted, sagittal magnetic resonance images of the spinal
cord; CV: Photomicrograph of a cresyl violet stained mid-sagittal section
through the lesion site; NF: Neurofilament stained mid-sagittal section
through the lesion site; Outcome (units): Functional outcome in locomotor
capacity (rat; BBB score) and ASIA lower limb motor score (human) and electrophysiological
outcome of motor evoked potentials (MEP: latency (ms) and amplitude (mV)
and somatosensory evoked potentials (SSEP: latency (ms) and amplitude (mV).
MRI reflects a similar pattern of tissue damage as seen in histology
and is paralleled by the functional and electrophysiological results reflecting
the degree of tissue loss.
Magnification histology: 40x; Scale bar rats = 0.7 mm; Scale bar
humans = 2 cm
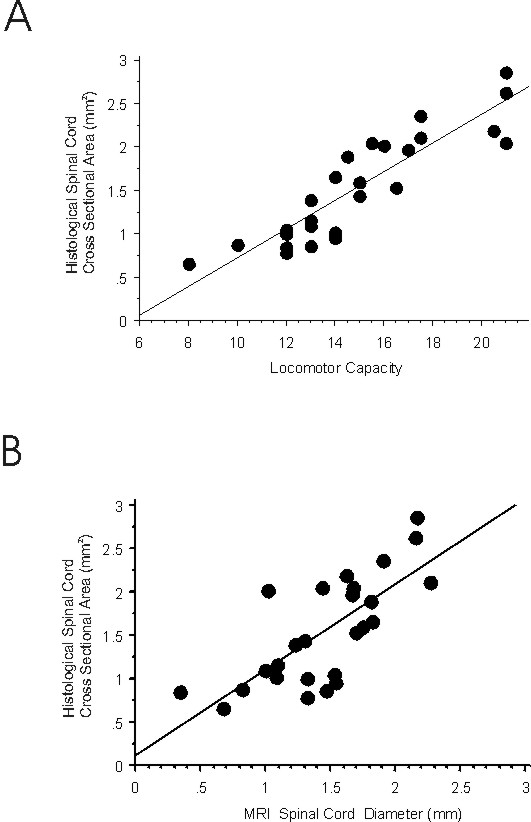
Figure 3: Correlations of MRI parameters with histological parameters of the same sample of injured spinal cords: A. In vivo MRI lesion size is closely correlated to lesion size as determined histologically post-mortem; B. Spinal cord diameter assessed in MRI is closely correlated to spinal cord cross sectional area in histology.
Summary and Conclusion
1. In rats and humans, electrophysiological measures correlate with
the outcome of motor function as assessed in rats by open field locomotion
and in humans by ASIA lower limb motor scores.
2. In morphological assessments in rats, we found signal enhancement
in MRI in areas that corresponded to post-trauma cavitation as seen in
histology of the same spinal cords.
3. Motor function in rats correlates with the morphological quantification.
4. MRI can also give valuable and accurate information about the extent
of a spinal cord lesion in patients where usually no other morphological
parameters are available.
5. The data infer that motor function and anatomy correlate in man
also.
6. Despite many physiological and anatomical differences between rat
and man, the sensitivity of techniques suggests an analogous relationship
between these parameters and comparable sensitivity of the techniques applied
in both species
Acknowledgments:
We thank Dr. K. Schenker (Bruker Medical, Spectrospin AG, Fällanden)
and Dr. J. Hodler (Orthopedic University Hospital Balgrist, Zurich) for
the valuable support in magnetic resonance imaging and Dr. R. Dürr
for preparing the analysis software. This study was supported by grants
of the Swiss National Science Foundation (NFP 38, grant No 4038-043918.95),
by the International Research Institute for Paraplegia (grant No P38/97)
and by the International Spinal Research Trust (Guildford, GB).
References
1. Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 1995; 12(1): 1-21.
2. Constantini S, Young W. The effects of methylprednisolone and the ganglioside GM1 on acute spinal cord injury in rats. J Neurosurg 1994; 80(1): 97-111.
3. Curt A, Dietz V. Ambulatory capacity in spinal cord injury: significance
of somatosensory evoked potentials and ASIA protocol in predicting outcome.
Arch phys Med Rehabil 1997; 78: 39-43.
4. Curt A, Keck ME, Dietz V. Functional outcome following spinal cord
injury: significance of motor-evoked potentials and ASIA scores. Arch Phys
Med Rehabil 1998; 79: 81-6.
5. Ditunno JF, Young W, Donovan WH, Creasey G. The international standards
booklet for neurological and functional classification of spinal cord injury.
Paraplegia 1994; 32: 70-80.
6. Gruner JA. A monitored contusion model of spinal cord injury in
the rat. J Neurotrauma 1992; 9(2): 123-28.
| Discussion Board | Previous Page | Your Poster Session |