Invited Symposium: Hypertension III: Flow-Induced Vascular Remodeling
| INABIS '98 Home Page | Your Session | Symposia & Poster Sessions | Plenary Sessions | Exhibitors' Foyer | Personal Itinerary | New Search |
Materials and Methods
Rat mesenteric blood flow model All experimental protocols used in this research project comply strictly with the guidelines of the Institutional Animal Care and Use Committee (IACUC). Three groups of 10 male Wistar rats (300 g) were anesthetized with Na+-pentobarbital (60 mg/kg IM; Anpro Pharmaceutical, Arcadia, CA), given 10,000 units penicillin G potassium IM (Marsam Pharmaceutical, Cherry Hill, NJ), and the surgical level of anesthesia maintained via supplemental Na+-pentobarbital. Abdomens were shaved and cleaned with a topical antiseptic/antimicrobial agent. Using sterile technique, a midline laparotomy was made and the cecum, small intestine, and supporting mesenteric membranes and vasculature exteriorized. The fourth, fifth, and sixth ileal arteries proximal to the ileocecal junction were isolated, and arterial ligations were made using sterile 10-0 Ethilon monofilament nylon suture (Ethicon, Inc., Somerville, NJ) as can be seen in Figure 1. The cecum, intestines, and mesentery were gently placed back inside the peritoneal cavity, the abdominal wall sutured with sterile 4-0 plain gut (Ethicon, Inc.), and the skin closed with sterile surgical clips. Animals were allowed to recover completely, placed back in the animal care facility, and given water and standard rat chow ad libitum for 1, 3, or 7 days.
 Click to enlarge
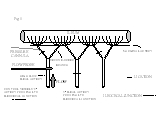
Fig.1: Rat Mesenteric Blood Flow Model indicating general mesenteric arterial arrangement, sites of ligation, and placement of pressure cannula and flow probe. This diagram depicts the model used for both acute hemodynamic measurements and chronic morphological assessments. In the chronic studies, the proximal branch of the high flow sixth ileal artery was ligated. Results showed significantly increased blood flows in both the ileal and second-order arteries, with a significantly elevated calculated wall shear rate for the high flow branching artery. No significant changes in mean carotid nor local mesenteric arterial pressures were detected. This model allows for same animal control and high flow vessels. Control vessels were obtained from the ninth ileal artery and second-order branch proximal to the ileocecal junction and are not included in the illustration.
Click to enlarge
Fig.1: Rat Mesenteric Blood Flow Model indicating general mesenteric arterial arrangement, sites of ligation, and placement of pressure cannula and flow probe. This diagram depicts the model used for both acute hemodynamic measurements and chronic morphological assessments. In the chronic studies, the proximal branch of the high flow sixth ileal artery was ligated. Results showed significantly increased blood flows in both the ileal and second-order arteries, with a significantly elevated calculated wall shear rate for the high flow branching artery. No significant changes in mean carotid nor local mesenteric arterial pressures were detected. This model allows for same animal control and high flow vessels. Control vessels were obtained from the ninth ileal artery and second-order branch proximal to the ileocecal junction and are not included in the illustration.
After the appropriate number of days, rats were re-anesthetized and mean arterial pressure (MAP) taken through tail artery cannulation for purposes of obtaining an established pressure for perfusion-fixation. A medial laparotomy was performed followed closely by a pneumothorax as the means for sacrifice. The intestines, mesenteric vasculature, and right kidney were perfusion-fixed at approximate MAP with a warmed vasodilator mixture [100 µM Na+ nitroprusside (Sigma, St. Louis, MO), 100 µM verapamil hydrochloride (Sigma), 100 µM papaverine hydrochloride (Aldrich Chemical Co., Milwaukee, WI), in normal saline] followed by warmed 10% buffered formalin phosphate (Fisher Scientific, Fair Lawn, NJ). The mesenteric vasculature was removed from the animal and the high flow and control arteries isolated and extracted. All tissues remained in 10% buffered formalin phosphate for approximately 4 hours before processing.
Hemodynamic Parameter Measurements In a duplicate series of experiments hemodynamic parameters were measured before and initially after creation of the model by arterial ligation. The left common carotid artery was cannulated to measure MAP. Following a medial laparotomy and surgical protocol as described above, a perivascular ultrasonic flow probe (0.5V probe and Model 206 Flowmeter, Transonics Systems Inc., Ithaca, NY) was placed on the sixth ileal artery proximal to the ileocecal junction (Fig. 1). The proximal second-order branch off the fourth ileal artery proximal to the ileocecal junction was ligated. In 6 animals, the proximal branch of the sixth ileal artery was cannulated retrogradely for the purpose of determining if creation of the model altered arterial pressure in the experimental mesenteric arteries. The cannula was constructed from 31 gauge MicroFil tubing (World Precision Instruments, Sarasota, FL) sealed in PE 10 and PE 50 tubing. The fifth ileal artery proximal to the ileocecal junction was then ligated. Measurements of blood flow, arterial pressure, and internal diameters of the sixth ileal artery and its distal second-order branch were made prior to and after each cannulation or ligation as described above. Ileal artery blood flow measurements were corrected for zero offsets, and measurements of internal diameters were made through videomicroscopy. Before accepting each pressure measurement, the stopcock connecting the cannula to the pressure transducer was opened to the atmosphere to verify that the cannula was patent as demonstrated by the entrance of blood into the cannula. Finally, vessels were completely relaxed through topical administration of a dilator cocktail (10-4 M adenosine, 10-5 M Na+ nitroprusside), with subsequent measurement of flow, pressure, and diameters. Average wall shear rate was calculated from these data using the formula: WSR = (4Q)/(pi*r3) where Q is blood flow (ml/sec) and r is the vessel radius (cm).
Tissue and Slide Preparation and Morphological Analysis Fixed tissues were processed in graded alcohols, paraffin-embedded, and stored at room temperature. Four micron sections were cut using a rotary microtome (American Optical 820, Buffalo, NY) and placed on precleaned Superfrost/Plus microscope slides (Fisher Scientific). For morphological analysis, slides were deparaffinized and rehydrated in graded alcohols and stained with toluidine blue for 45 seconds at 37 °C. The slides were then dehydrated in graded alcohols and xylene and coverslipped. For in situ hybridization experiments, control and treatment vessels were placed on the same slide to reduce variation. Microscopic measurement and quantitation was performed using JAVA Video Image Analysis Software System (Jandel Scientific, Jandel Corp.). Between 3 and 6 cross-sections per vessel were measured for morphometric analysis.
Extracellular Connective Tissue Staining Tissue cross-sections were stained for extracellular connective tissue using a modification of the protocol included for Masson's Trichrome staining (Sigma). After slides were stained, microscopic evaluation was performed using Image-1 (Universal Imaging Corporation, West Chester, PA) linked to a Sony DXC-151 CCD camera (Sony, Japan) and Nikon Optiphot-2 microscope (Nikon, Japan). Percent connective tissue in the vessel medial wall cross-sections was achieved using quantitative stereology. An average of 8 measurements were made per vessel cross-section for percent extracellular connective tissue.
Immunohistochemistry for Proliferating cell nuclear antigen (PCNA) Standard immunohistochemistry protocols were used to stain for PCNA and eNOS. Endogenous peroxidase activity was quenched followed by 5% normal goat serum as a blocking agent (Vector). For PCNS immunostaining, a mouse monoclonal anti-PCNA clone PC-10 (Boehringer Mannheim Corp., Indianapolis, IN) was used in a 1:400 dilution for 2 hours at room temperature. For eNOS immunostaining, a mouse affinity-purified monoclonal IgG-1 (Transduction Laboratories, Lexington, KY) was used in a 1:1000 dilution overnight at 4 °C. The secondary antibody, biotinylated goat anti-mouse IgG (Vector), was used in a 1:400 dilution for 30 minutes. Slides were stained using the Vectastain Elite Avidin-Biotinylated Complex immunoperoxidase system (Vector) with a diaminobenzidine tetrahydrochloride (DAB; Sigma) chromagen. Slides were counter-stained, dehydrated, and coverslipped. Positive and negative nuclei were counted in the medial layer, and between 3 and 5 cross-sections per vessel were analyzed. The right kidney served as a positive control for DNA replication, and the negative control slides received only the secondary antibody.
In situ hybridization The cDNA for human PDGF-A chain was obtained from Collins and co-workers (17). A Sac II-Sal I restriction fragment from 544-913 bp was subcloned into pBluescript SK+. Using restriction endonuclease digestion Sal I, T3 polymerase transcribed a 368 bp sense strand, while Sty I and T7 polymerase gave a 321 bp anti-sense strand. All enzymes and buffers for 35S-CTP riboprobe syntheses were purchased from Promega Corporation (Madison, WI). Sense and anti-sense riboprobes were transcribed in the presence of 90 µCi 35S-CTP (Dupont de Nemours, Boston, MA) in a standard 20 µl transcription reaction containing 1 µg linearized cDNA and the appropriate RNA polymerase T3 or T7. Transcription proceeded for 2 hours at 37 °C. Radionucleotide incorporation was measured using PEI cellulose-F chromatography paper (EM Industries, Inc., Gibbstown, NJ) and phosphorimager analysis (Molecular Dynamics). Samples were purified, the DNA eluted, and the probe precipitated in cold 100% ethanol and stored at -70 °C until use. Prior to use, the precipitate was resuspended in 10 µl formamide and 10 µl 20 mM DTT and radioactivity was measured to ensure at least 106 CPM/µl.
The in situ hybridization methods were primarily based on those described by Wilcox (18) for formalin-fixed, paraffin-embedded tissues. Slides were deparaffinized and washed in 0.5 x SSC, deproteinated with 5 µg/ml proteinase K (Amresco, Solon, Ohio), and rinsed in PBS. Slides were post-fixed in 4% paraformaldehyde (Sigma), rinsed, and covered with 200 µl prehybridization stock for 2 hours at approximately 40 °C. The tissues were then incubated with 100 µl hybridization solution containing radioactive probe at 106 CPM/slide in the humidity chamber (Isotemp Incubator, Fisher Scientific) at 55 °C overnight. The next day slides were washed in a low stringency 2xSSC solution and treated to a 40 µg/ml RNAse A solution (Sigma). Slides were washed in the low stringency solution, followed by 1-hour washes (x4) in a high stringency solution at 55 °C. Slides were rinsed, serially dehydrated in graded alcohols, and air dried overnight.
The following day, slides were dipped in Kodak NTB-2 emulsion (Eastman Kodak Co.) and placed in a sealed incubator box containing desiccant. After 10 weeks in emulsion, slides were developed and counterstained according to standard procedures. Autoradiographic evaluation of specific and background silver grains was performed using the Image-1 system linked to a Sony DXC-151 CCD camera (Tokyo, Japan) and Nikon Optiphot-2 microscope (Tokyo, Japan). Data from duplicate samples (>3) were averaged, background was subtracted from specific signal, and mean values for sense were subtracted from those for anti-sense to provide an overall densitometric value. Data were presented as number of grains per 50 µm2 of tissue for wall area, and number of grains per 15 µm luminal perimeter for the endothelium.
Statistical analysis Data were stored and analyzed on personal computers using Excel (Microsoft version 5.0), Instat 2 (Graphpad Software, version 2.02), and Sigma Plot (Jandel Scientific; version 2.0). Flow, pressure, and WSR data were analyzed using a repeated measures Analysis of Variance (ANOVA) with post-hoc Student-Newman-Keuls analysis. Morphological and endothelial count data were analyzed using a two-way ANOVA with post-hoc Bonferroni-corrected paired and unpaired T-tests. All other data including those from densitometric in situ hybridization analyses were analyzed using a two-tailed paired Student's T-test. The null hypothesis was rejected at an alpha of 0.05. Unless otherwise specified, data were presented as mean±SEM.
| <= Introduction | MATERIALS & METHODS | Results => |
| Discussion Board | Next Page | Your Symposium |